Research Article
Determination of the Toxicity Effect of Sorbitan Esters Surfactants Group on Biological Membrane
Kerman Pharmaceutics Research Centre, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
P. Khazaeli
Department of Pharmaceutics, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
S. Mirzaei
Kerman Pharmaceutics Research Centre, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
F. Sharififar
Kerman Pharmaceutics Research Centre, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
S. Nasrollahosaiani
Kerman Pharmaceutics Research Centre, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran










Miloni Reply
Dear Sir,
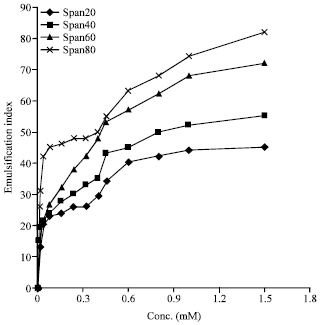
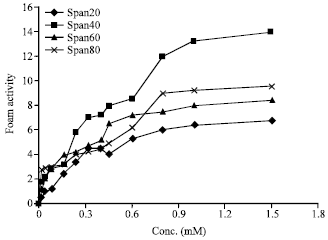
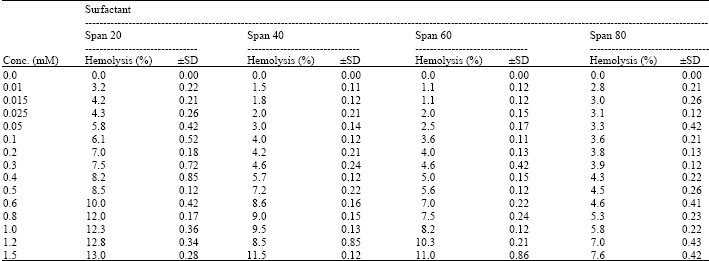
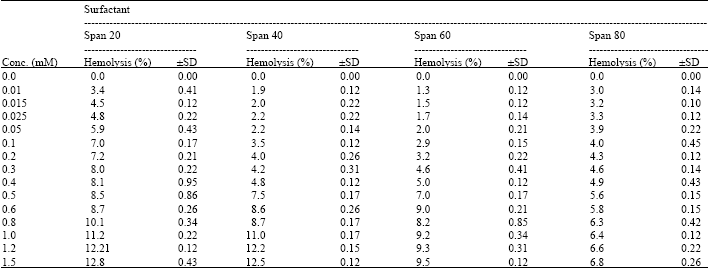
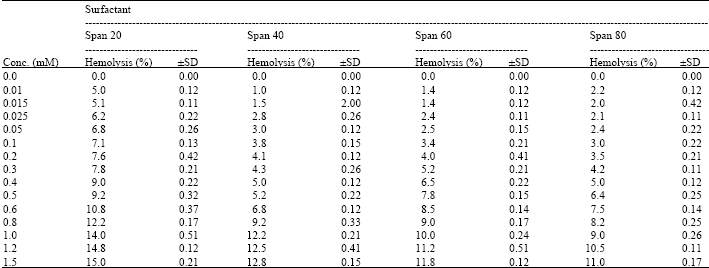
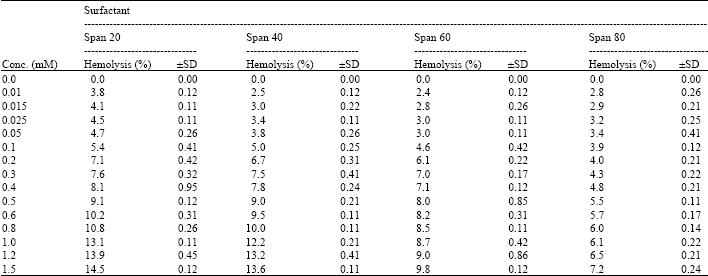
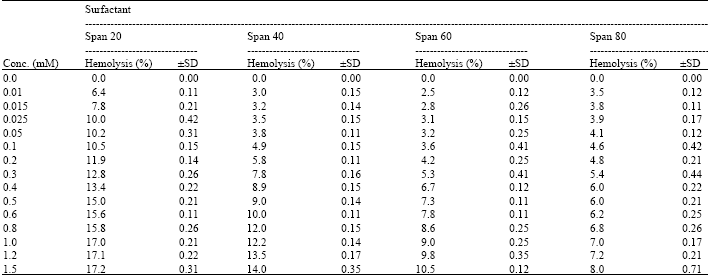
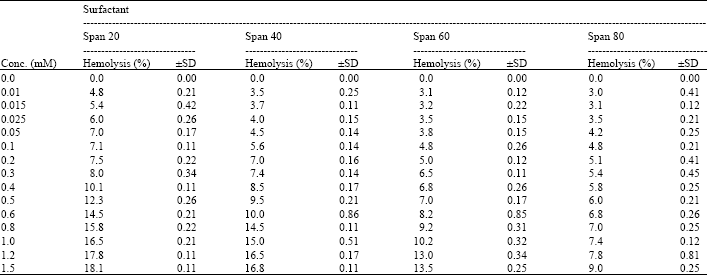
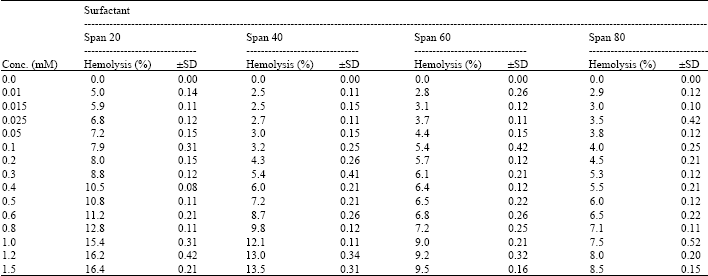
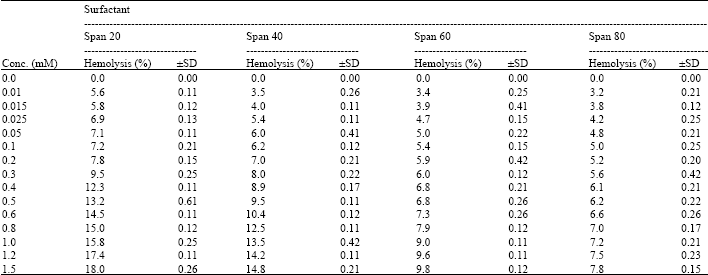
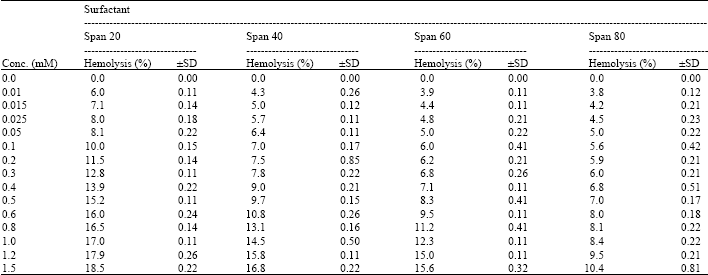
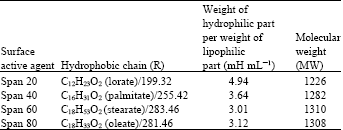
I want to do a similar study but have a few questions to ask. For the emulsification index you have considered and studied the E24 at various concentrations of surfactants. However, you have not mentioned as to in which system you made these concentrations. Could you please highlight on the solvent system used. Or did you simply use water? Also is it important to study the E24 index by varying the concentrations or pH of the emulsion system?? Also how did you decide the varying concentrations of the surfactants for the toxicity study? Was it based simply on CMC? Should the concentration be considered logarithmically from the CMC value?
I would be grateful if you could answer my questions.
Regards,
Miloni