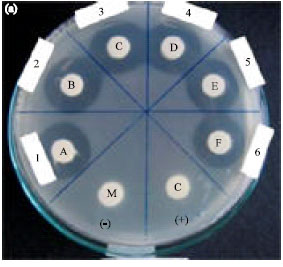
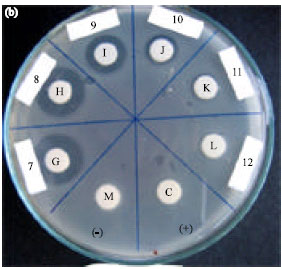
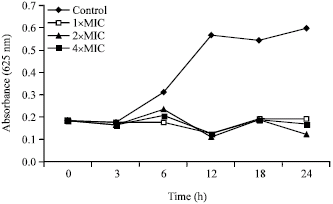
Research Article
In vitro Antibacterial Activity of Methanol Extract of A Sponge, Geodia sp. Against Oxytetracycline-Resistant Vibrio harveyi and its Toxicity
Fish Diseases Laboratory, Department of Fisheries, Faculty of Agriculture, Gadjah Mada University, Jl. Flora, Bulaksumur, Yogyakarta 55281, Indonesia
Trijoko
Laboratory of Animal Taxonomy, Faculty of Biology, Gadjah Mada University, Jl. Teknika Selatan, Yogyakarta 55281, Indonesia
E.P. Setyowati
Laboratory of Biological Pharmacy, Faculty of Pharmacy, Gadjah Mada University, Sekip Utara, Yogyakarta 55281, Indonesia
H.H. Anshory
Fish Diseases Laboratory, Department of Fisheries, Faculty of Agriculture, Gadjah Mada University, Jl. Flora, Bulaksumur, Yogyakarta 55281, Indonesia