Research Article
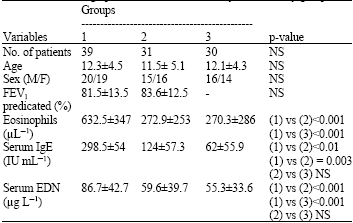
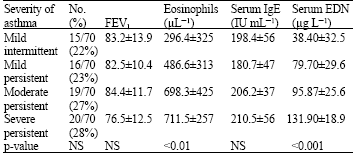
Eosinophil-Derived Neurotoxin Versus Immunoglobulin E as Biomarkers for Evaluation of Bronchial Asthma
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
A. El-Wan
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
Y. Kamel
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
M. Nabih
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
H. Mahmoud
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
M. Almasry
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt
H. Hussein
66 Mohi El-Deen, Abo El-Ezz Street, Dokki, Guiza, Egypt