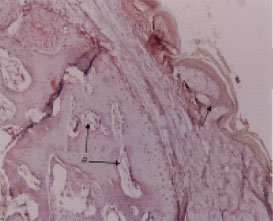
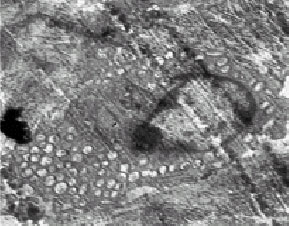
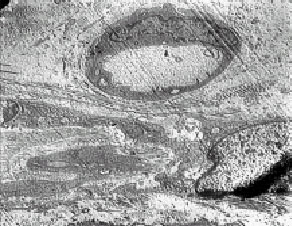
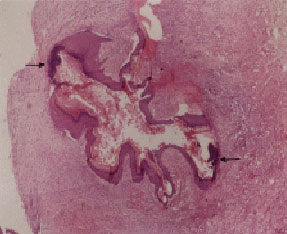
Research Article
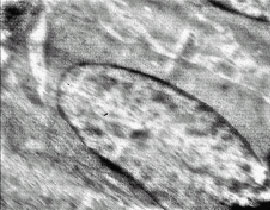
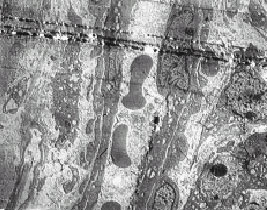
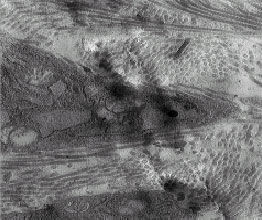
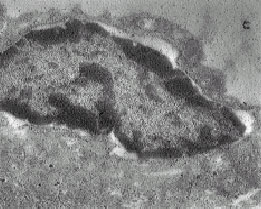
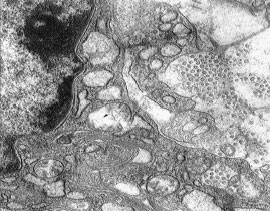
The Ultra Structural Study of Blastema in Pinna Tissues of Rabbits with Transmission Electron Microscope
Department of Biology, School of Sciences, Ferdowsi University of Mashhad, Iran
Fatemeh Naseri
Department of Biology, School of Sciences, Ferdowsi University of Mashhad, Iran
Masoumeh Kheirabadi
Department of Biology, School of Sciences, Ferdowsi University of Mashhad, Iran
Sakineh Babaie
Department of Biology, School of Sciences, Islamic Azad University, North Tehran Branch, Iran
Fereshteh Sadeghie Shakib
Department of Biology, School of Sciences, Ferdowsi University of Mashhad, Iran
Mahnaz Azarniya
Department of Biology, School of Sciences, Tarbiat Moalem University, Tehran, Iran