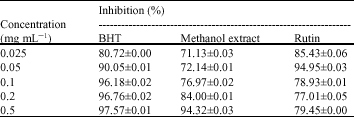
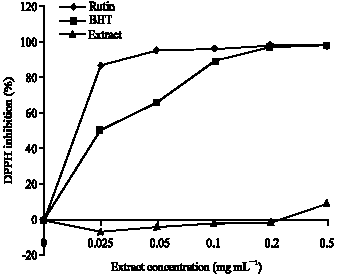
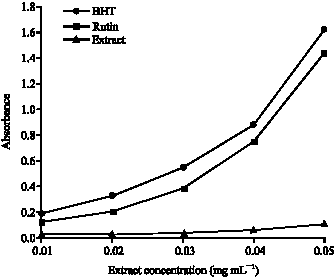
Research Article
Total Phenolic Content and Free Radical Scavenging Activity of Malva parviflora L. (Malvaceae)
Department of Botany, University of Fort Hare, Alice, South Africa
O.M. Aboyade
Department of Botany, University of Fort Hare, Alice, South Africa
M.O. Sofidiya
Department of Pharmacognosy, University of Lagos, Akoka, Nigeria