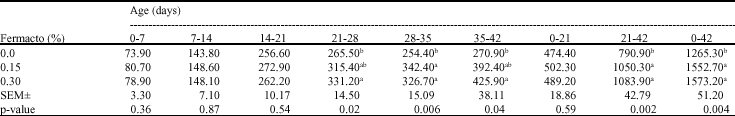
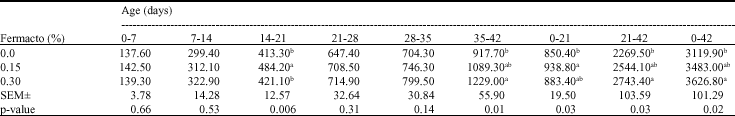
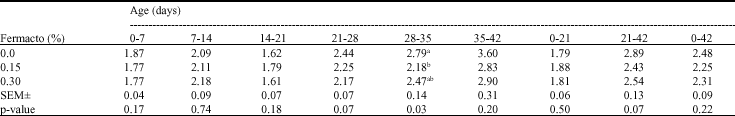
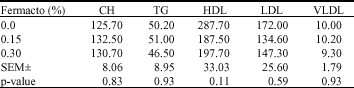
Research Article
Effects of Diet Supplementation of Aspergillus Meal Prebiotic (Fermacto®) on Efficiency, Serum Lipids and Immunity Responses of Broiler Chickens
Department of Animal Science, Faculty of Agriculture, Ferdowsi University of Mashhad, P.O. Box 91775-1163, Mashhad, Iran
H. Kermanshahi
Department of Animal Science, Faculty of Agriculture, Ferdowsi University of Mashhad, P.O. Box 91775-1163, Mashhad, Iran