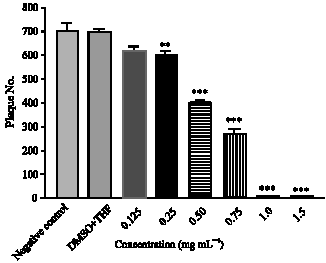
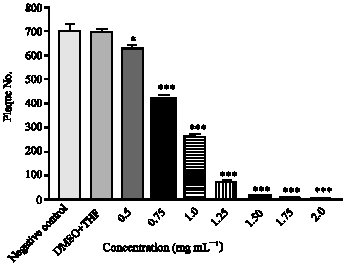
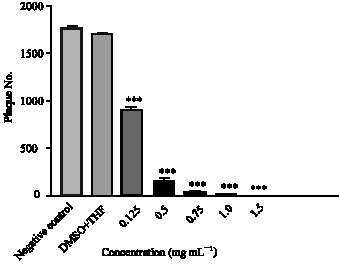
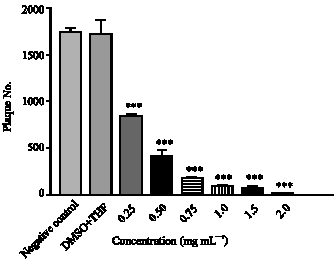
Research Article
Antiviral Activity of Euphorbia helioscopia Extract
Pharmaceutical Research Center, Bu-Ali Research Institute and School of Pharmacy, Mashhad University of Medical Sciences, P.O. Box 91775-1365, Mashhad, Iran
J. Behravan
Biotechnology Research Center, Bu-Ali Research Institute and School of Pharmacy,Mashhad University of Medical Sciences, P.O. Box 91775-1365, Mashhad, Iran
M. Arab
Pharmaceutical Research Center, Bu-Ali Research Institute and School of Pharmacy, Mashhad University of Medical Sciences, P.O. Box 91775-1365, Mashhad, Iran
S. Amel Farzad
Pharmaceutical Research Center, Bu-Ali Research Institute and School of Pharmacy, Mashhad University of Medical Sciences, P.O. Box 91775-1365, Mashhad, Iran