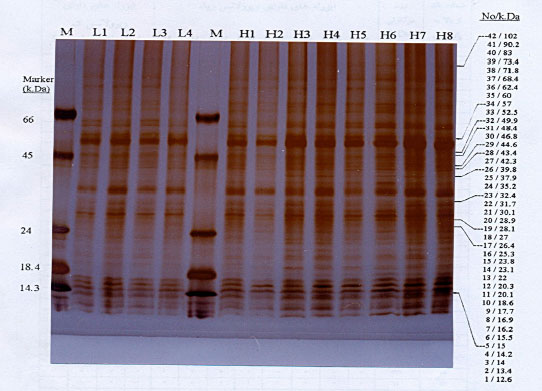
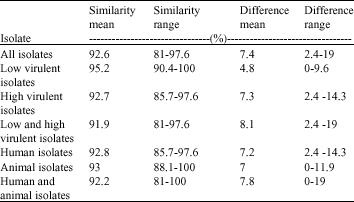
Research Article
Characterization of the Similarity of Protein Patterns and Virulence of Clinical Candida albicans Isolates
Mycology Research Center, Faculty of Veterinary Medicine, University of Tehran, Tehran, Iran
M. Riazipour
Department of Mycology, Faculty of Medicine, University of Imam Hossein, Tehran, Iran
H. Shokri
Mycology Research Center, Faculty of Veterinary Medicine, University of Tehran, Tehran, Iran
M.L. Mousavi
Department of Biochemistry, Faculty of Medicine, University of Imam Hossein, Tehran, Iran
M. Mahmoudi
Department of Epidemiology, Faculty of Medical Sciences, Tehran, Iran