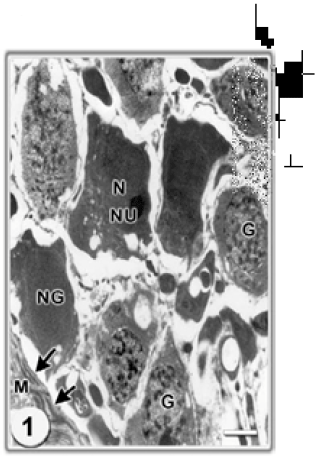
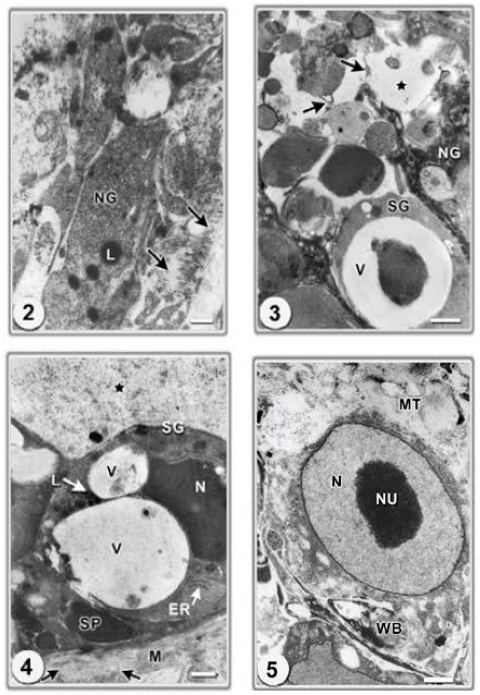
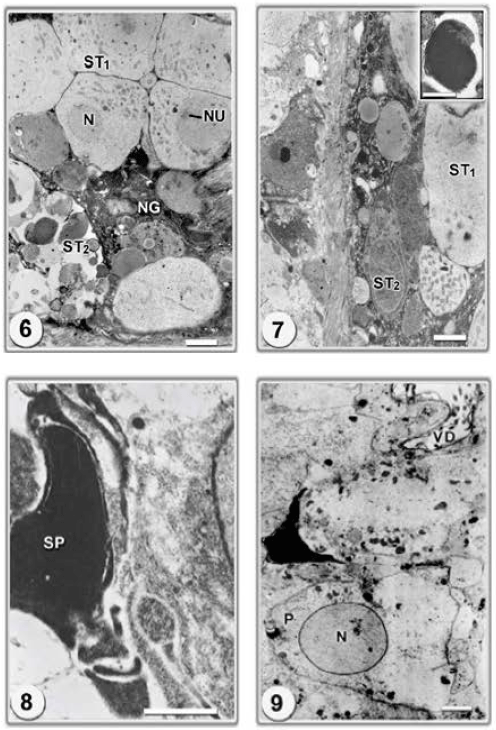
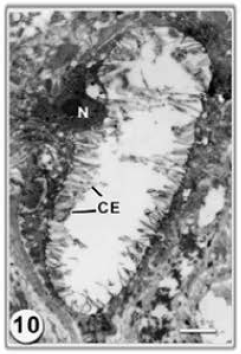
Research Article
Ultrastructural Alterations in Testis and Gastrodermis of Schistosoma mansoni Due to Treatment of Infected Mice with the New Rhodanine Derivative Ro-354
Department of Zoology, Faculty of Science, Ain Shams University, Abbasia, Cairo, Egypt