Research Article
Some Biochemical Properties of Catalase from Kohlrabi (Brassica oleracea gongylodes)
Department of Biochemistry, Faculty of Veterinary Medicine, University of Tabriz, Tabriz, Iran
INTRODUCTION
Catalase (EC 1.11.1.6), is one of the most potent catalysts known. The reactions it catalyses are crucial to life. Catalase catalyses conversion of hydrogen peroxide, a powerful and potentially harmful oxidizing agent, to water and molecular oxygen. Catalase also uses hydrogen peroxide to oxidize toxins including phenols, formic acid, formaldehyde and alcohols (Scandalios et al., 1997; Chaudiere and Ferrari-lliou, 1999; Karra-Chaabouni et al., 2003; Oztürk et al., 2007). It is one of the first enzymes to be purified and crystallized and has gained a lot of attention in recent years because of its link to cancer, diabetes and aging in humans and animals (Bloch et al., 2007; Melov et al., 2000; Preston et al., 2001; Turdi et al., 2007). In plants, catalase scavenges H2O2 generated during mitochondrial electron transport, β-oxidation of the fatty acids and most importantly photorespiratory oxidation (Scandalios et al., 1997).
Accumulating evidence indicates that catalase plays an important role in plant defense, aging and senescence (Mura et al., 2007; Conrath et al., 1995). Catalase has been characterized from many prokaryote and eukaryote organisms so far. Catalase has been reported to exist in multiple forms in many higher plants, such as saffron, mustard, spinach, cotton, wheat, sunflower, maize, castor bean, Pepper, loblolly pine and tobacco (Havir and McHale, 1987; Mullen and Gifford, 1993; Garcia et al., 2000; Keyhani et al., 2002; Lee and An, 2005).
Kohlrabi (Brassica oleracea gongylodes) is a low, stout cultivar of the cabbage that will grow almost anywhere and used as a food and also as a traditional medicine. The plant is carefully described in botanical books, but there is no available information on the basic aspects of its physiology and biochemistry. Our objective in this study was to investigate and characterize catalase activity in kohlrabi.
MATERIALS AND METHODS
Plant materials: This study was performed during June to October 2007. Kohlrabi was collected during summer from the North-west of Iran (Tabriz) and stored at 4 °C. For the preparation of the crude extract, 100 g of Kohlrabi bulbs was finely chopped and homogenized with 100 mL of 0.05 M phosphate buffer, pH 7.0 containing 0.1% polyvinylpyrrolidone (for stabilizing the extract) in a waring blender for 5 min. The homogenate rapidly filtered through a layer of cheesecloth and centrifuged at 20,000 g for 30 min at 4 °C. A clear, transparent supernatant termed crude extract was obtained and used for our studies. Protein concentrations were determined by the method of Lowry et al. (1951) using crystalline bovine serum albumin as standard.
Catalase activity assay: Catalase activity was measured by following the dismutation of H2O2 spectrophotometrically using an extinction coefficient for H2O2 at 240 nm of 27 M-1 cm-1 (Obinger et al., 1997). Assays were carried out at room temperature (~22-25 °C) with an UNICO UV-2100 PC spectrophotometer. One hundred microliter extract (965 μg protein mL-1) were added to 0.1 M citrate-phosphate-borate buffer, at given pH, containing H2O2 in final concentration of 0.5 to 38 mM (for Km, Vmax determinations) or 10 mM (for all other assays) the reaction mixture total volume was 3 mL. Reaction velocity was computed from linear slopes of absorbance-time curve (Wong et al., 1971). One unit of catalase activity was defined as the amount of enzyme necessary for reducing 1 μmol of H2O2 per min.
Thermal stability assay: Thermal stability of kohlrabi catalases were studied by incubating aliquots of extract at various temperatures (25-70 °C) up to 30 min in a thermostatic water bath and measuring their activity at room temperature after brief cooling in ice. The incubation was carried out in sealed vials to prevent change of volume of the sample and hence, the enzyme concentration due to evaporation. The activity was measured at 25 °C, was taken as 100% and activities which were measured (35-70 °C) were compared with the activity measured at 25 °C.
Activity staining for catalase: Catalase activity in native PAGE gels (7%) was determined as described by Woodbury et al. (1971). Gels were incubated in 0.003% H2O2 for 10 min and developed in a 1% (w/v) FeCl3 and 1% K3Fe(CN6) (w/v)) solution for 10 min.
Chemicals: Chemicals for electrophoresis and protein assay reagent were purchased from Sigma Chem. Co. All the other chemicals used in this research were obtained from Merck and were of reagent grade.
RESULTS AND DISCUSSION
Kinetic studies for identification of various types of catalases in kohlrabi bulbs were based on the following criteria as suggested by Schulz (1994) and Fullbrook (1996): (i) variation in activity as a function of pH; (ii) variation in activity as a function of substrate concentration (Km and Vmax); (iii) effect of inhibitors on activity (differential effects of azide and cyanide); (iv) effect of temperature on activity.
Figure 1 shows the H2O2 dismutation by kohlrabi bulbs extract in the presence of 10 mM H2O2 at different pH, ranging from 3.5 to 11.0 expressed as units of enzyme per mg extract protein. Three peaks were found, respectively at pH 4.5, 6.5 and 10. As suggested by Fullbrook (1996) the presence of various pH optima indicates the presence of distinct enzymes. Table 1 shows the Km, Vmax and catalytic efficiency (Km/Vmax, calculated per mg extract protein) at the respective pHs. The Km, Vmax and catalytic efficiency were different for all three pHs examined. Their distinctive kinetic properties suggested possible different metabolic roles for each enzyme.
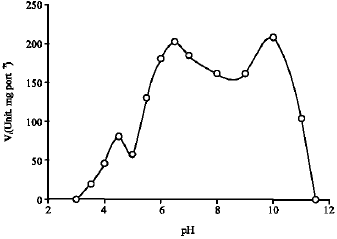 | |
| Fig. 1: | pH dependency of catalase activity in kohlrabi bulbs |
Effect of substrate concentration (hydrogen peroxide) on activity alteration at three pH optima showed that only catalase activity at pH 4.5 was inhibited by hydrogen peroxide at concentration above 5 mM. At pH 4.5, the maximum rate was 299.27 μM min-1 for a substrate concentration of 5 mM; thereafter, substrate inhibition was observed and for 15 mM H2O2, the activity was reduced to 44.89 μM min-1 (results not shown).
The effect of two inhibitors, namely azide (NaN3) and cyanide (NaCN) on catalase activity in kohlrabi bulbs extract were also studied (Fig. 2, Table 1).
As shown in Table 1, catalase activity at pH 4.5 in comparison to other two pH optima has a highest and lowest sensitivity to azide and cyanide, respectively. Order of sensitivity of catalase activity to azide at three pH optima was pH 4.5>6.5>10, with IC50 of 10, 75 and 1000 μM, respectively. IC50 value was found for cyanide at pH 4.5 was 1200 times greater than to azide, showing that catalase activity at pH 4.5 was remarkably insensitive to cyanide in comparison to other two pH optima. The results confirming the presence of at least three distinctive isoenzymes of catalase in kohlrabi bulbs extract as suggested by the three pH optima.
The thermal stability of the catalase activity in kohlrabi bulbs extract, presented as the residual percentage activity was shown in Fig. 3.
As shown in this Fig. 3 after heating the extract at 70 °C for 30 min, catalase activity lost about 40, 60 and 75% of the original activity at pH 4.5, 10 and 6.5, respectively. When the extract was incubated at specified temperature (25-70 °C) for various length of time (5-30 min) then tested at room temperature a decrease in catalase activity was found at temperature above 50, 60 and 70 °C for pHs of 6.5, 10 and 4.5, respectively (results not shown). Therefore, the order of thermostability of three
| Table 1: | Kinetic parameters and sensitivity to azide and cyanide of the catalase activity in kohlrabi bulbs extract at three pH optima |
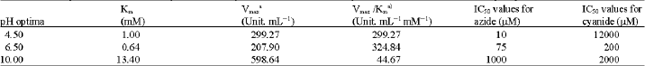 | |
| aCalculated per mg extract protein | |
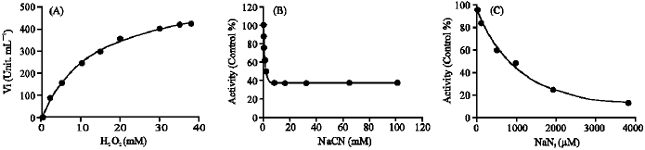 | |
| Fig. 2: | (A) Variation in velocity of the reactions catalyzed by kohlrabi catalase as a function of substrate concentration, at pH 10. Vmax and Km were determined by fitting the data to Michaelis-Menten equation by sigmaplot 9. (B) Inhibition by cyanide against kohlrabi catalase at pH 10. (C) Inhibition by azide against kohlrabi catalase at pH 10 |
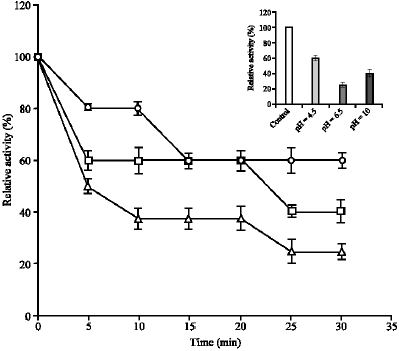 | |
| Fig. 3: | Thermal stability of kohlrabi catalases at pH 4.5 (o), 6.5 (Δ) and 10 (). Extract was incubated for various time intervals (5-30 min) at 70 °C and then tested at room temperature, at given pH, after brief cooling in ice. The activity was measured at 25 °C was taken as 100%. Inset: Relative activity (% control) of kohlrabi catalases after heating the extract at 70 °C for 30 min |
catalases isoenzymes from kohlrabi bulbs extract at three pH optima was pH 4.5>10>6.5.
The presence of three isoenzymes was confirmed by when the extract was submitted to non-denaturating PAGE, followed by activity staining for catalase (Lane A in Fig. 4).
Three distinct bonds corresponding to apparent molecular weights of 308,000, 285,000 and 258,000 were revealed after staining for catalase activity. After preincubation of the extract with 100 μM azide, only two bands (bands 2 and 3) were visualized on the gel (Lane B in Fig. 4). This result suggested that band 1 probably
 |
| Fig. 4: | Non-denaturing PAGE of extract stained for catalase activity. Lane A was loaded with 20 μL extract (20 μg protein) and lane B was loaded with 20 μL extract (20 μg protein)+azide (100 μM). Band 1, 2 and 3 corresponded to apparent molecular weights of 308,000, 285,000 and 258,000, respectively |
could be corresponded to the catalase isoenzyme active at pH 4.5. Since catalase activity at pH 4.5 was more sensitive to azide (IC50 = 10 μM) in comparison to other two pH optima (IC50 = 75 μM for pH 6.5, IC50 = 1000 μM for pH 10).
Present results suggested at least three types of isoenzymes of catalase in kohlrabi bulbs extract with high apparent molecular weights of 308, 285 and 258 kDa. Such these high molecular mass are similar to three catalases isoenzyme of dormant saffron (Crocus sativus L.) corms with apparent molecular weights of 323, 295 and 268 kDa and Catalase isolated from leaves of the halophytic plant Mesembryanthemum crystallinum with high apparent molecular mass of about 320 kDa in previous studies (Keyhani et al., 2002; Niewiadomska and Miszalski, 2007). High molecular weight of catalases also was found in some fungal and bacterial species: 320 kDa in Neurospora crassa (Jacob and Orme-Johnson, 1979), 337 kDa in Escherichia coli (Clairborne and Fridovich, 1979) and 260 kDa in Methanobrevibacter arboriphilus (Shima et al., 2001). Whilst, most of the catalases of higher plants are characterized by molecular masses of about 240 kDa (Scandalios et al., 1997). Optimum pH for catalases activity in kohlrabi bulbs extract was found between 4.5 and 10. Keyhani et al. (2002) showed that three isoforms of catalase in dormant saffron corms was at its optimum pH between 6 and 11. Several researchers have found similar results for plant catalases and indicated an optimum pH between 5 and 10 (Yoruk et al., 2005; Garcia et al., 2000; Dincer and Aydemir, 2001). Unlike animals, plants have evolved multiple isoforms of catalase that are differentially regulated in various environmental stresses (Lee and An, 2005). The presence of multiple isoforms in kohlrabi bulbs extract with distinctive kinetic properties and different sensitivity to temperature and inhibitors (azide and cyanide) suggest that each of isoform plays a different role. Further experiments should clarify the overall roles of each of isoforms in environmental stresses.
ACKNOWLEDGMENT
This research was supported in part by Grant number 861016 from Research Affairs of the University of Tabriz.