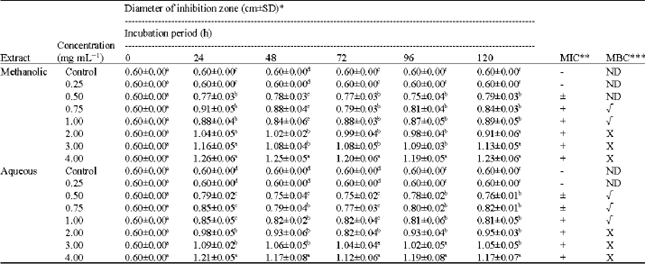
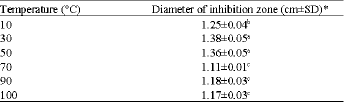
Research Article
Comparative Study on the in vitro Antibacterial Efficacy of Aqueous and Methanolic Extracts of Quercus infectoria Gall`s Against Cellulosimicrobium cellulans
M. Dirnahayu and C.K.N. Che Ku Nurshaira Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
Y. Nurhafiza
M. Dirnahayu and C.K.N. Che Ku Nurshaira Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
A.A. Nor Azwady
M. Dirnahayu and C.K.N. Che Ku Nurshaira Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
E. Nor Dalilah
M. Dirnahayu and C.K.N. Che Ku Nurshaira Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia