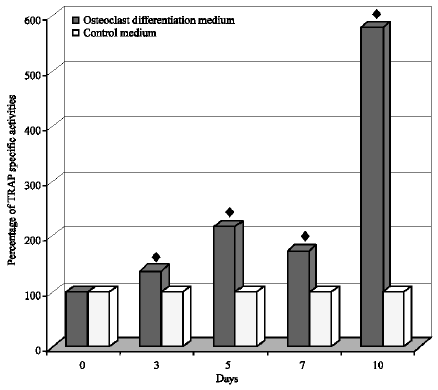
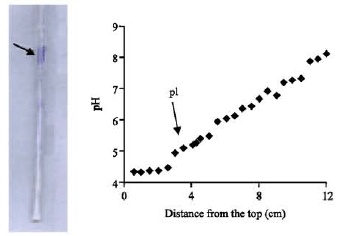
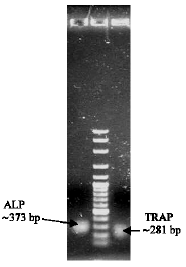
Research Article
Osteoclast and Osteoblast Development of Mus musculus Haemopoietic Mononucleated Cells
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Selangor, Malaysia
Z.A. Shahrul Hisham
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Selangor, Malaysia
M.A.W. Rohaya
Department of Orthodontics, Faculty of Dentistry, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
S. Sahidan
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Selangor, Malaysia
Z.A. Zaidah
Department of Microbiology, Faculty of Applied Sciences, Universiti Teknologi Mara, Selangor, Malaysia