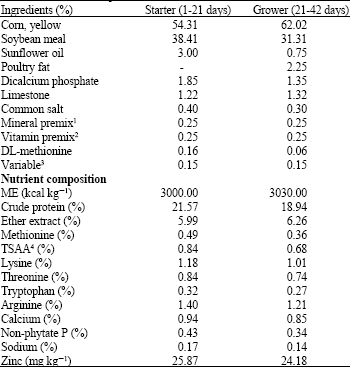
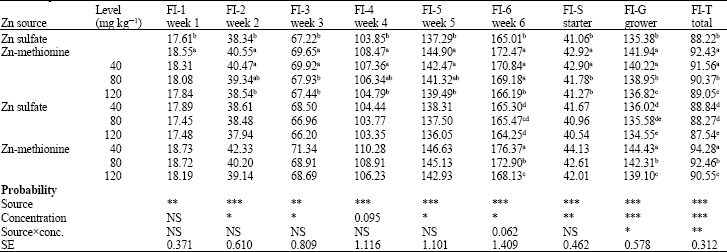
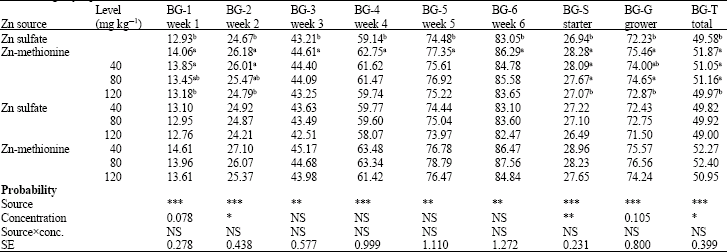
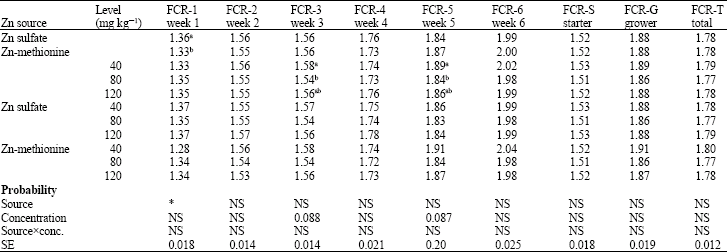
Research Article
The Influence of Dietary Zinc-Methionine Substitution for Zinc Sulfate on Broiler Chick Performance
Department of Animal Science, Faculty of Agriculture, Ferdowsi University of Mashhad, Mashhad 91775-1163, Iran
H. Nassiri Moghaddam
Department of Animal Science, Faculty of Agriculture, Ferdowsi University of Mashhad, Mashhad 91775-1163, Iran
A. Rezaei
Department of Immunology, Faculty of Medicine, Isfahan University of Medical Sciences, Isfahan, Iran
A.R. Haghparast
Department of Veterinary Immunology, College of Veterinary Sciences, Ferdowsi University of Mashhad, Mashhad, Iran