Research Article
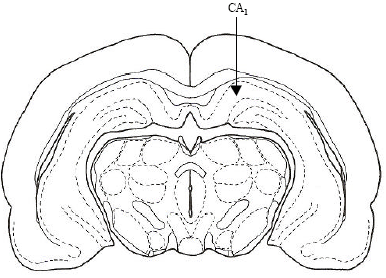
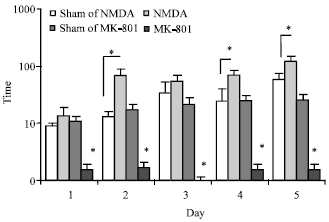
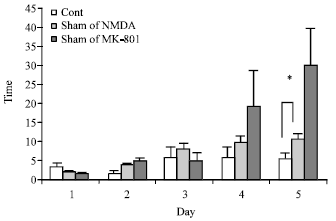
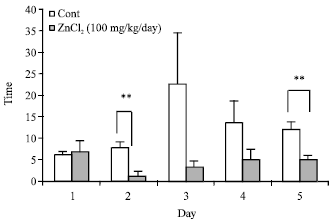
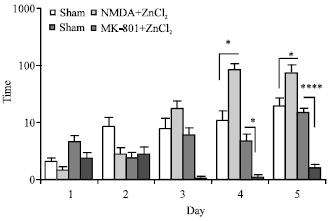
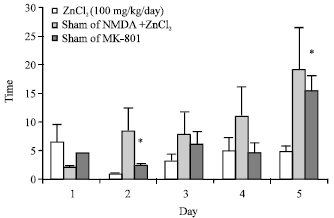
The Role of CA1 Hippocampus NMDA Receptors to Passive Avoidance Task in Presence and Absence of ZnCl2 in Adult Male Rats
Department of Biology, School of Science, Shahid Chamran University, Ahwaz, Iran
Z. Valizadeh
Department of Biology, School of Science, Shahid Chamran University, Ahwaz, Iran
G. H.A. Parham
Department of Statistic, School of Mathematic, Shahid Chamran University, Ahwaz, Iran