Research Article
Effect of Narasin and Dietary Protein Source on Performance of Broiler
Department of Animal Science, Faculty of Agriculture, University of Kurdistan, Sanandaj, Kurdistan, Iran
Narasin is a mono-valent polyether ionophore (Greek: ioon = going; phoreo = transport) that is a fermentation products of Streptomyces aureofaciens, which is used in aid in the prevention of coccidiosis and necrotic enteritis in broiler chicks (Branuius, 1985; Brennan et al., 2003; Chapman, 2001; Guneratne and Gard, 1991; Johansson et al., 2004).
Narasin has been by far the second most commonly used antimicrobial in commercial poultry production, not only used for their anticoccidial effect, but also as growth promoters in Eimeria-free environment, due to their effect in improving feed conversion efficiency (Branuius, 1985; Waldenstedt and Elwinger, 1995; Chapman, 2001; Johansson et al., 2004). Brennan et al. (2001) showed that narasin is effective in the preventions of necrotic enteritis (NE) in broiler chicks. Brennan et al. (2003) also demonstrated the effectiveness of narasin fed alone and in combination with bacitracin methylene disalicylate in the management of necrotic enteritis using a Clostridium perfringens inoculum challenge model. There is no doubt that the removal of antibiotic growth promoters from animal feeds has resulted in a much higher rate of NE in broiler flocks. Future plans to ban the use of coccidiostats will make the situation further complicated. The poultry industry will have to learn to cope with the new conditions, as they are learning to handle welfare demands.
Fish meal is the most important conventional animal protein source for poultry in most developing countries. It is fed to farm animals to improve productivity, preserve health and welfare and to reduce dependence on antibiotics and other drugs (Babu et al., 2005). It has been well established that the beneficial effects of fish meal on broiler performances are mediated mainly via improvement of palatability and hence broiler daily feed intake (Karimi, 2006). In addition both fish oil and fish meal provides a concentrated source of long-chain n-3 fatty acids, eicosapentaenoic acid (C20:5, n-3, EPA) and docosahexaenoic acid (C22:6, n-3,DHA), which has been showed to reduced the adverse affects of coccidiosis on growth and reduced gut lesion scores in coccidiosis challenged chicks in the absence of coccidiostats. These claimed effects of fish lipids on chicks immune system is mainly postulated to mediate by moderating the immune reaction to disease challenged and improving specific immunity (Bartov and Jensen, 1980; Pike, 1999; Wiesenfeld, 2005). It has been showed that when chicks were challenged with coccidiosis in the absence of coccidiostats, incorporation of omega-3 fatty acids in the diet reduced the adverse effects on growth and reduced gut lesion scores (Allen and Danforth, 1998). Increasing demand, high costs and uncertain availability of fish meal, together with risk factors associated with disease from animal protein sources, have resulted in nutritionists studying alternative sources for inclusion into the diets of poultry (Babu et al., 2005).
Whilst the potential of narasin as a valuable coccidiostat and effective product in prevention of NE in broiler feeding is not in doubt (Watkins and Bafudo, 1993; Conway et al., 2001), little research has been done to determine whether animal protein sources with a potential contributing to the outbreaks of clinical NE [21], such as fishmeal influence the broilers response to narasin, in a coccidial free environment. Hence, to the best of our knowledge, there is less information on the effects of narasin and none on the interactions between protein source and narasin on broiler performance, the objective of the study in this report was to examine the performance response of broiler chickens to diets supplemented with different levels of narasin in combination with dietary protein sources.
The present study was carried out at the Animal Science Department of University of Kurdistan, Kurdistan, Iran, during January to February 2006.
Three hundred and fifty two day-old, strait run, Hubbard broiler chicks, were randomly allocated to four dietary treatments, each replicated four times (22 chicks per pen) in a completely randomized design in a 2x2 factorial arrangements. The chicks were housed in floor pens (1.2x1.5 m) using wood shavings as bedding material throughout the experiment. Lighting was continuous for the first day post-hatching, after which a 23L: 1D lighting schedule was maintained for the duration of the experiment. Temperature was maintained between 32 and 34 °C at the beginning of the rearing period and was gradually decreased every 2 to 3 day to 22 °C at the end of rearing period. Chicks were provided free access to feed and water during the experimental period. Care and management of the chicks were in accordance with commercial guidelines. The conditions and standards of care employed in this study were approved by the ethical committee for animal experiments of University of Kurdistan.
Dietary treatments: The corn-soybean meal-based starter (0-20 day) diets were formulated to meet or exceeded the requirements (NRC, 1994) for all nutrients. Experimental diets were formulated to contain 0 or 50 g kg-1 of fish meal. The ionophoric anticoccidial agent, narasin supplement (with 0 or 60 mg kg-1 of the diet) was added directly to the diets (Table 1).
Measurements: The experiment was conducted for 20 days. Birds were weighed as a group on arrival. At 10 and 20 day, all birds were weighed and feed intake was determined. Feed conversion was calculated after adjusting for daily mortality. Average body weight, daily gain, feed intake and feed to gain ratio (FCR) were calculated for each period and for the overall experiment.
| Table 1: | Composition (%) and calculated analysis of basal diets |
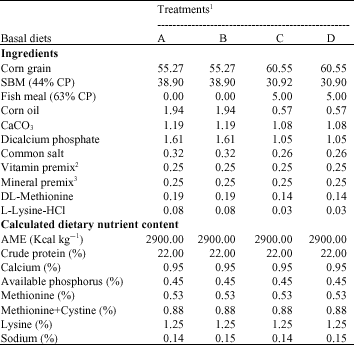 | |
| 1Treatments: A = Control (no fish meal, no narasin); B = No fish meal, 60 mg kg-1 of narasin; C = 5% fish meal, no narasin; D = 5% fish meal and 60 mg kg-1 of narasin 2Provides per kg of diet: Vit. A, 9000 IU; Vit. D3, 2000 IU; Vit. E, 18 IU; Menadion, 2 mg; Thiamine, 1.8 mg; Riboflavin, 6.6 mg; Niacin, 30 mg; Pyridoxin, 3 mg; Vit B12, 15 mcg; D-Pantothenic acid, 100 mg; Folic acid, 1 mg; Biotin, 0.1 mg; Choline chloride, 500 mg; Antioxidant, 100 mg 3Provides per kg of diet: Manganese, 100 mg; Zinc, 84.7 mg; Iron, 50 mg; Copper, 10 mg; Iodine, 1 mg; Se, 0.2 mg | |
Data were analyzed according to General Linear Model (GLM) procedure of SAS (2001) as a Completely Randomized Design (CRD) in a factorial arrangement. Significant differences among treatments were determined at p<0.05 by Duncan`s new multiple range tests. Pen was used as the experimental unit.
During the experimental period mortality was within acceptable level (less than 2%) and was not related to dietary treatments. The influences of different dietary treatments on broiler performance are shown in Table 2.
The results of this experiment showed that broiler liveweight at 10 day and average daily gain during the 0-10 and 11-20 day periods were not significantly influenced by 50 g kg-1 fishmeal inclusion to the diets (p>0.05). However, the results indicated that chick body weight at 20 day and average daily gain through experiment were significantly improved (about 6%) with fishmeal supplementation to the diets (p<0.05). The results also showed that daily feed intake was significantly (p<0.05) increased by fishmeal level during the 11-20 and 0-20 day periods, but no such effect was noted between 0-10 day (p>0.05). The results of this experiment on the fish meal effects on broiler performances is consistent with previous reports (El Boushy and van der Poel, 1994; Karimi, 2006) in that the beneficial effects of fishmeal on broiler performances are mediated mainly via improvements of diet palatability, which consequently resulted in greater nutrient supply for chick growth. Many feed formulators ensure a minimum incorporation of fish meal in poultry diets, based on practical experience and experimental results indicating that this level of fish meal produces increases in meat production and better feed utilization compared with diets not containing fish meal. These minima are justified when the income from the increased performance exceeds the cost of incorporating these minimum levels in the diet. In addition it has been shown that dietary fish meal may enhance the immune response in poultry, which prove useful in infections in which cell-mediated immunity plays a role (Babu, 2005).
| Table 2: | The effect of different fish meal level (%) and narasin supplementation on performance in broiler chicks (Mean ± SD) |
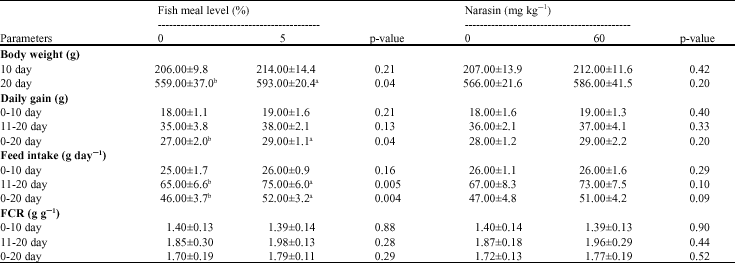 | |
| a, b: Mean values within a row and under each main effects with no common superscripts differ significantly (p<0.05) | |
The results of this experiment also showed that supplementation of experimental diets with coccidiostatic compound, narasin had no significant impact on broiler performance (p>0.05). The lack of significant effects of narasin on broiler performance in this study reconfirms the previous finding that anticoccidial drugs have no effects on broiler performance in a relatively coccidia-free environment. It must be noted that differences in exposure to coccidial challenge by poultry might be a factor in the growth response to polyether ionophorous compounds (Conway et al., 1999). Waldenstedt et al. (1999) reported that inoculated broiler chicks with a mixture of chicken Eimeria isolates had a 10% lower live weight than un-inoculated chickens and the performance of un-inoculated birds was similar to that of inoculated birds treated with narasin. Watson et al. (2005) also showed that broiler chick`s daily gain, average feed intake and gain: feed ratio was reduced by coccidial inoculation. It should be borne in mind that nearly all anticoccidial drugs have no positive effects on final body weight or even cause slight growth depression, even at the recommended levels in the absence of coccidial exposure (Braunius, 1985; Radu et al., 1987; Pesti et al., 1999).
In addition, lack of growth promoter effects of narasin in this study might be partially due to the lower prevalence of C. perfringens in birds given narasin in this study, since the birds were not inoculated with Clostridium perfringens inoculums and kept in well disinfected environment. Brennan et al. (2003) demonstrated that Bacitracin Methylene Disalicylate (BMD) and narasin, fed alone and in combination, are effective in reducing morbidity, mortality, NE lesion scores and suppression of growth and feed efficiency associated with NE among broiler chickens challenged with C. perfringens. Johansson et al. (2004) in a comparative study of different antimicrobial drugs also demonstrated that the narasin is still a potent antimicrobial agent against C. perfringens and development of resistance is apparently slow.
The interaction between fishmeal and narasin supplementation on chicks performance (Table 3) was not significant (p>0.05). The growth and intake depressing effects of anticoccidials was the main interest in this study, since it was hypothesized that these negative effects may be partially overcome via the growth and feed intake stimulating effects of fish meal. Given the lack of response to narasin fed in this experiment, it is not surprising that there was no interaction with the inclusion rate of fishmeal. One potential reason for this may be related to the type of anticoccidial drug used in this study and the marginal differences in protein and amino acids provided with our different dietary treatments. Bartov and Jensen(1980) reported that monensin induced growth depression was greater in birds fed on animal protein containing diets than in those fed a corn-soybean diet, but Pesti et al. (1999) showed that there were no significant interactions between protein source and semduramicin on growth (all plant protein or 12% of the protein content from animal origin). In conclusion, the results of the present experiment showed that the beneficial effects of fishmeal on broiler performance becomes most evident during the later stages of the early growth period of broilers, mainly via stimulation of feed intake. The results indicated that under condition of coccidial free environment, Narasin had no significant effect on broiler chick`s performance during starter period and did not result in any interaction with fish meal level.
| Table 3: | The interaction effects of fish meal levels (%) and narasin supplementation (with or without) on performance in broiler chicks (Mean ± SD) |
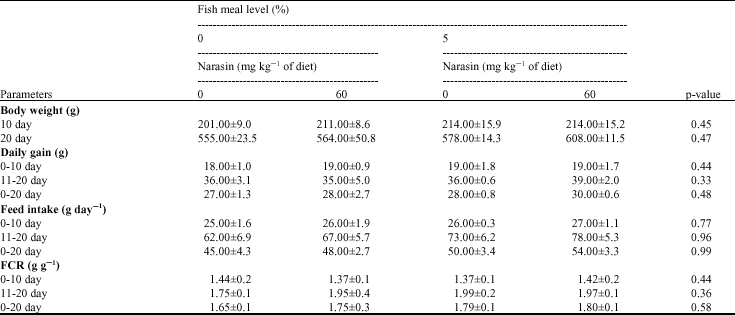 | |
| a, b: Mean values within a row with no common superscripts differ significantly (p<0.05) | |
We also gratefully acknowledge the valuable comments and suggestions of Dr. Michael Bedford (Zymetrics Inc., Marlborough, UK ) and Dr. A. Kamyab (Animal Science Res. Ctr., University of Missouri) during preparation of this article.