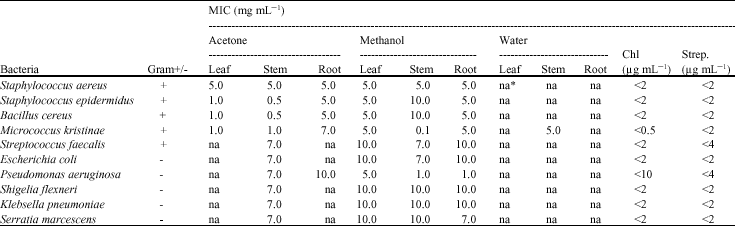
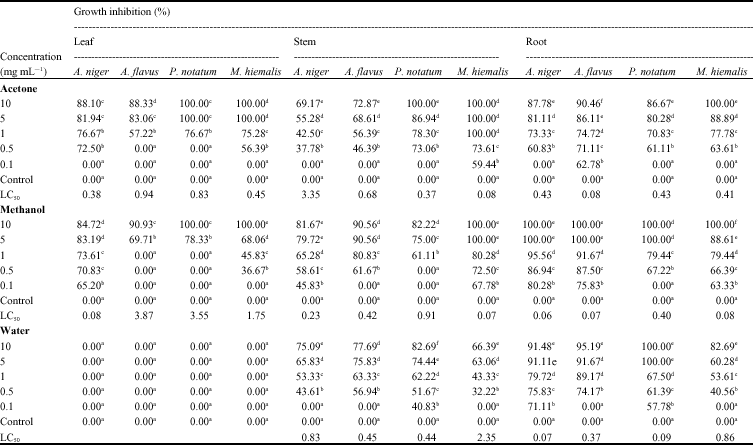
Research Article
Antimicrobial Activity of Extracts from Felicia muricata Thunb
Department of Botany, University of Fort Hare, Alice, South Africa
D.S. Grierson
Department of Botany, University of Fort Hare, Alice, South Africa
A.J. Afolayan
Department of Botany, University of Fort Hare, Alice, South Africa