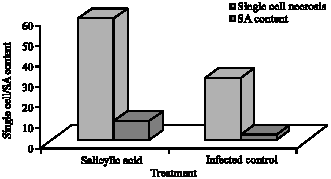
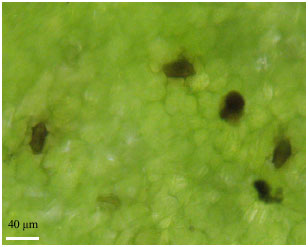
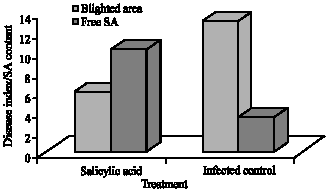
Research Article
Exogenous Applications of Salicylic Acid for Inducing Systemic Acquired Resistance Against Tomato Stem Canker Disease
Department of Plant Protection, College of Agriculture, Bu-Ali Sina University, Hamadan, Iran
M.J. Soleimani
Department of Plant Pathology, Michigan State University, East Lansing MI 48824, USA
H. Rouhani
Department of Plant Protection, College of Agriculture, Ferdosi University, Mashhad, Iran