Research Article
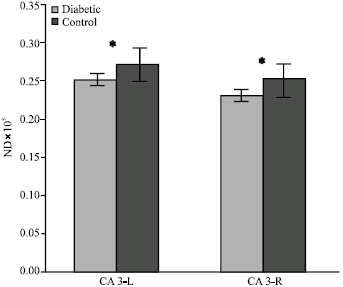
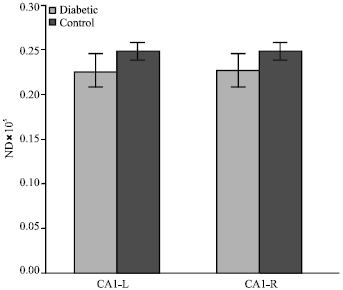
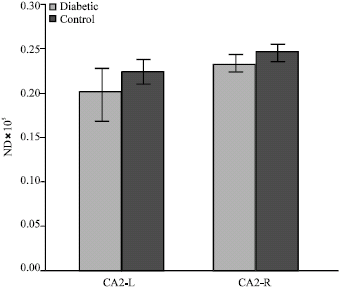
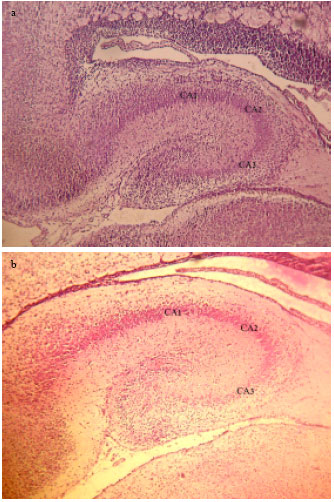
Effect of Maternal Diabetes on Hippocampus Neuronal Density in Neonatal Rats
Department of Biology, Faculty of Science, Islamic Azad University, Mashhad Branch, Mashhad, Iran
M.R. Khakzad
Medical University, Islamic Azad University, Mashhad Branch, Mashhad, Iran