Research Article
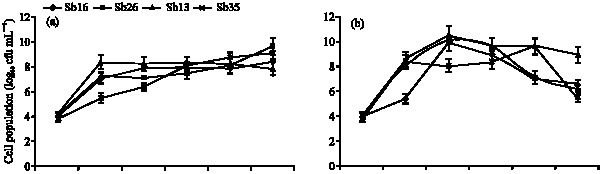
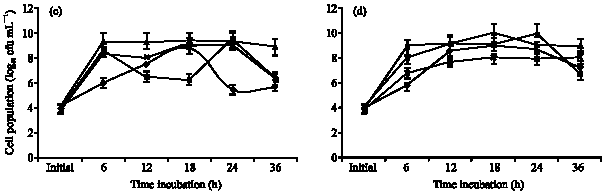
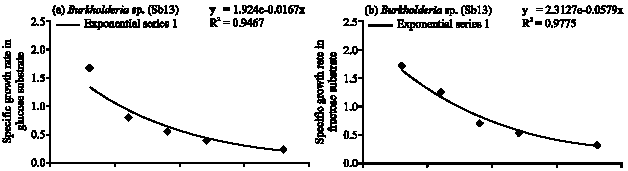
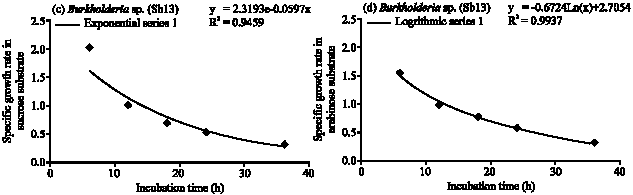
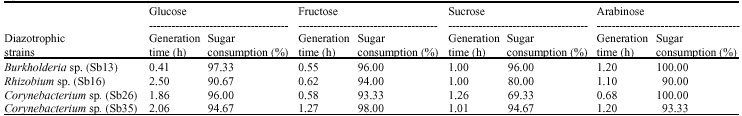
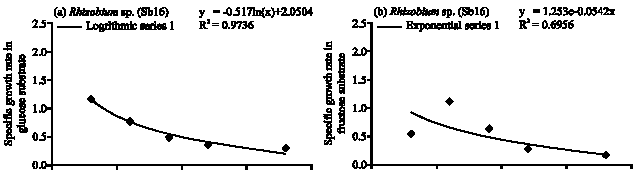
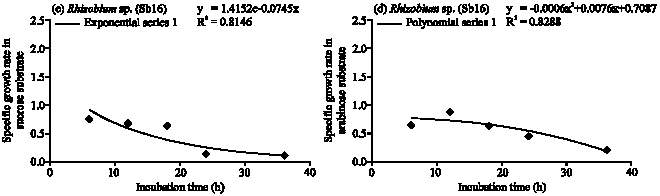
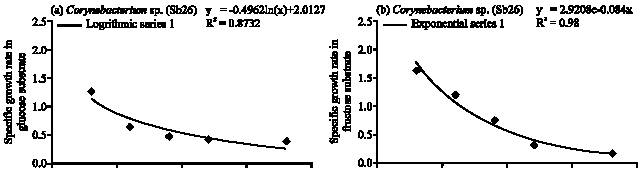
Specific Growth Rate and Carbon Sugar Consumption of Diazotrophs Isolated from Rice Rhizosphere
Department of Land Management, Universiti Putra Malaysia (UPM), 43400, Serdang, Selangor DE, Malaysia
O. Radziah
Department of Land Management, Universiti Putra Malaysia (UPM), 43400, Serdang, Selangor DE, Malaysia
M.S. Halimi
Department of Agricultural Technology,Universiti Putra Malaysia (UPM), 43400, Serdang, Selangor DE, Malaysia
Z.H. Shamsuddin
Department of Land Management, Universiti Putra Malaysia (UPM), 43400, Serdang, Selangor DE, Malaysia
I.M. Razi
Department of Crop Science, Universiti Putra Malaysia (UPM), 43400, Serdang, Selangor DE, Malaysia