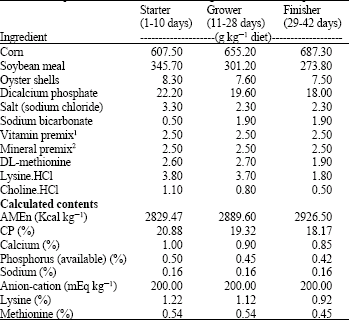
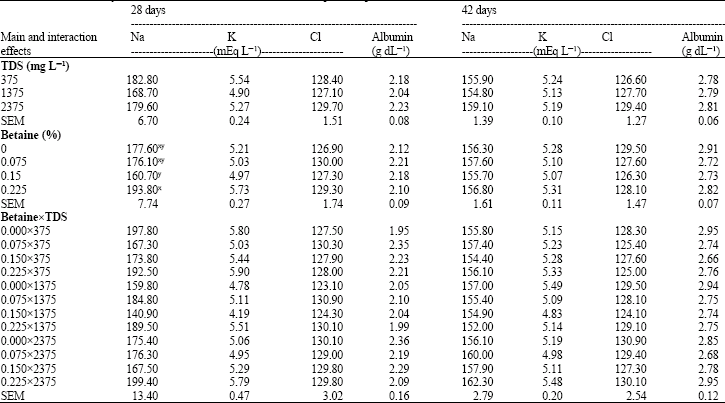
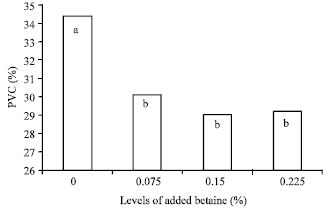
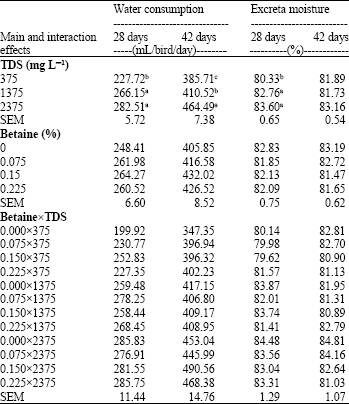
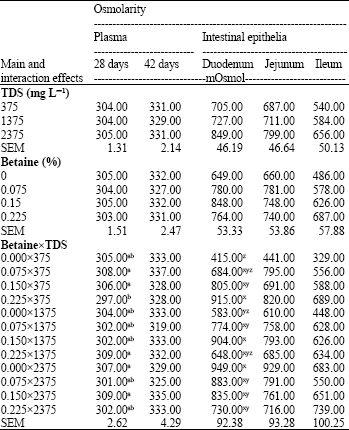
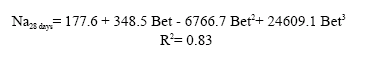Research Article
The Effect of Betaine on Water Salinity Tolerance in Broiler Chicks
Department of Animal Science, Faculty of Agriculture, University of Tehran, Karaj, Iran
Mojtaba Zaghari
Department of Animal Science, Faculty of Agriculture, University of Tehran, Karaj, Iran
Mahmood Shivazad
Department of Animal Science, Faculty of Agriculture, University of Tehran, Karaj, Iran