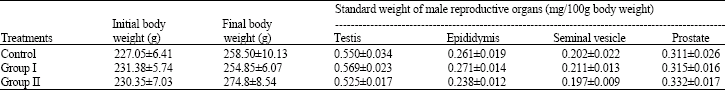
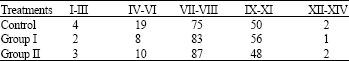Research Article
Effects of Aqueous Extract of Anethum graveolens (L.) On Male Reproductive System of Rats
Department of Biology, College of Sciences, Shiraz University, Shiraz, Iran
Pahlavan Sara
Department of Biology, College of Sciences, Shiraz University, Shiraz, Iran