ABSTRACT
The present study was focused for investigation the effects of sublethal concentration of nickel exposure after three weeks on some physiological and gill structure alterations in the freshwater fish, Oreochromis niloticus. Physiological parameters such as serum sodium, chloride, osmolality, glucose, cholesterol, total protein, albumin, amylase, lipase, alanine aminotransferase and aspartate aminotransferase were chosen to evaluate the response of experimental animal to nickel intoxication. In comparison with control, serum sodium, chloride and osmolality values were decreased in nickel-exposed fish, while the levels of serum glucose, cholesterol, total protein, albumin, amylase, lipase, alanine aminotransferase and aspartate aminotransferase were significantly elevated. Nickel exposure induced some histological changes in fish gill structure. These changes included hyperplasia, hypertrophy, shortening of secondary lamellae and fusion of adjacent lamellae. The physiological and histological changes indicate that nickel is very hazardous pollutant. Moreover, the above mentioned severe alterations indicate that the fish, Oreochromis niloticus is appropriate species to act as a biological indicator of water pollution level.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jbs.2007.77.85
URL: https://scialert.net/abstract/?doi=jbs.2007.77.85
INTRODUCTION
Environmental contamination and exposure to heavy metals is a serious growing problem throughout the world. Heavy metals are taken into the body via inhalation, ingestion and skin absorption. If heavy metals enter and accumulate in body tissue faster than the body’s detoxification pathways can dispose of them, a gradual buildup of these toxins will occur. In general, heavy metals are systemic toxins with specific neurotoxic, nephrotoxic, hepatotoxic, fetotoxic and teratogenic effects. Heavy metals can directly influence behavior by impairing mental and neurological function, influencing neurotransmitter production and utilization and altering numerous metabolic body processes. Systems in which toxic metal elements can induce impairment and dysfunction include the blood and cardiovascular, eliminative pathways (colon, liver, kidneys, skin), endocrine (hormonal), energy production pathways, enzymatic, gastrointestinal, immune, nervous (central and peripheral), reproductive and urinary (ATSDR, 1996). The toxic effects of heavy metals on fish are multidirectional and manifested by numerous changes in the physiological and chemical processes of their body systems (Dimitrova et al., 1994).
Nickel is a very abundant element. In the environment, it is found primarily combined with oxygen (oxides) or sulfur (sulfides). It is found in all soils and is emitted from volcanos. Nickel has properties that make it very desirable for combining with other metals such as iron, copper, chromium and zinc to form alloys. These alloys have important uses such as in the making of metal coins and jewelry and in industry for making items such as valves and heat exchangers. Most nickel is used to make stainless steel. Nickel compounds are used for nickel plating, to colour ceramics, to make some batteries and as substances known as catalysts to increase the rate of chemical reactions. Nickel is released into the atmosphere during nickel mining and by industries that convert scrap or new nickel into alloys or nickel compounds or by industries that use nickel and its compounds. These industries may also discharge nickel in wastewater. Nickel is also released into the atmosphere by oil-burning power plants, coal-burning power plants and trash incinerators. The nickel that comes out of the stacks of the power plants is attached to small particles of dust that settle to the ground or are taken out of the air in rain. It will usually take many days for nickel to be removed from the air particularly if it is bound to very small particles. Acidic conditions render nickel more mobile in soil and may lead to seepage into groundwater. Exposure to nickel occurs through breathing air or smoking tobacco containing nickel, eating food containing nickel (the major source of exposure for most people), drinking water which contains nickel, handling coins and touching other metals containing nickel. However, the major sources of nickel exposure are tobacco smoke, auto exhaust, fertilizers, superphosphate, food processing, hydrogenated-fats-oils, industrial waste, stainless steel cookware, testing of nuclear devices, baking powder, combustion of fuel oil, dental work and bridges. The most common adverse health effect of nickel in humans is an allergic reaction. People can become sensitive to nickel when jewelry or other things containing this metal come into direct contact with the skin. Once a person is sensitized to nickel, further contact with it will produce a reaction. Symptoms include burning, itching, redness and bumps or other rashes. A rash may spread to other areas and last for weeks after exposure stops, but usually improves in about a week less frequently, some people who are sensitive to nickel have asthma attacks following exposure to nickel. Eye or lung effects, including chronic bronchitis and reduced lung function, have been observed in workers who inhaled large amounts of nickel. High exposure can cause cough, shortness of breath and fluid in the lungs, which is sometimes delayed for 1 to 2 days after exposure. Single high or repeated lower exposures may damage the lungs, with scarring of lung tissues and may cause damage to heart muscle, liver and/or kidney. Fumes from heated nickel can cause a pneumonia-like illness, with cough and shortness of breath. Higher exposures can cause a build-up of fluid in the lungs (pulmonary edema), a medical emergency, with severe shortness of breath. People who are not sensitive to nickel must ingest very large amounts to show adverse health effects. Workers who accidentally drank water containing very high levels of nickel (100,000 times more than in normal drinking water) had stomachaches and effects to their blood and kidneys. Exposure to Nickel can cause a sore or hole in the bone dividing the inner nose (septum). Nickel and certain nickel compounds may reasonably be anticipated to be carcinogens. Cancers of the lung, nasal sinus and throat have resulted when workers breathed dust containing high levels of nickel compounds while working in nickel refineries or nickel processing plants. There may be no safe level of exposure to carcinogens. Carcinogens may also have the potential for causing reproductive damage in humans. Nickel may damage the developing fetus. Nickel is required to maintain health in animals although large doses have harmful effects. Animal studies show that breathing high levels of nickel compounds may result in inflammation of the respiratory tract. Eating or drinking large amounts of nickel has been reported to cause lung disease in dogs and rats and to affect the stomach, blood, liver, kidneys, immune system and reproduction and development in rats and mice. When rats and mice breathed nickel compounds for a lifetime, nickel compounds that were hard to dissolve caused cancer, while a soluble nickel compound did not cause cancer. Nickel has been shown to cause lung cancer in animals. Nickel and its compounds have high acute and chronic toxicity to aquatic life. Insufficient data are available to evaluate or predict the effects of nickel and its compounds to plants, birds, or land animals. Acute toxic effects may include the death of animals, birds, or fish and death or low growth rate in plants. Water hardness affects nickel toxicity to aquatic organisms. The concentration of nickel and its compounds found in fish tissues is expected to be somewhat higher than the average concentration of nickel and its compounds in the water from which the fish was taken (ATSDR, 1996). Additionally, epidemiologic studies have shown that nickel compounds cause nasal and lung cancers in exposed workers (Grandjean et al., 1988; Grimsrud et al., 2003). Nickel-containing compounds induced tuomors and enhanced tuomorgenesis in various experimental animals and human cell culture via several different types of exposure (Damjanov et al., 1987; Kasprzak et al., 1983; Hartwig et al., 1994; Kang et al., 2003). DNA-protein cross-links and chromosomal aberrations were observed in mammalian cells in culture to which nickel compounds were added (Patierno et al., 1985; Sen et al., 1987; Ren et al., 2001). In addition, nickel caused oxidative damage to isolated DNA and chromatin, possibly due to the formation of reactive oxygen species (Kasprzak, 1991; Kang et al., 2003; Kaur and Dani, 2003). Recently, several reports showed that nickel-induced various toxicological, physiological and histopathological alterations in different animal species (Pane et al., 2003; Bersenyi et al., 2004; Brix et al., 2004; Doreswamy et al., 2004; Hoang et al., 2004; Gupta et al., 2006).
The present study aimed to investigate the effect of nickel exposure on the freshwater fish, Oreochromis niloticus. Physiological parameters such as serum sodium, chloride, osmolality, glucose, cholesterol, total protein, albumin, amylase, lipase, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were chosen to evaluate the response of experimental animal to nickel intoxication. In addition, the toxic effects of nickel exposure on gills were evaluated histopathologically. Moreover, gill histopathology is an essential tool in fish pathology, physiology and aquatic toxicology. Both environmental factors and the presence of pathogens may lead to significant changes to gill structure. These changes may provide vital clues in an investigation of fish morbidity and mortality. Finally, the increasing emphasis on the assessment and monitoring of estuarine ecosystems has highlighted the need to deploy appropriate biological indices for these locations. Fish diseases and histopathology, with a broad range of causes, are increasingly being used as indicators of environmental stress since they provide a definite biological end-point of historical exposure (Stentiford et al., 2003).
MATERIALS AND METHODS
Animals: The experiments were conducted with adult Oreochromis niloticus of both sexes, which purchased from a local farm. To recover from transport conditions, fish were maintained for one week in several 200 L glass aquaria. The same temperature conditions (26±1°C) and pH (7.2±0.1) were maintained throughout the maintenance and experimental period. In addition, the acclimation water was changed daily to minimize accumulation of excretory products. The experimental animals were regularly fed with dry pellets (Nutrafin, Taiwan). The composition of these pellets as follows:
Ingredients: Fish meal, plankton, shrimp meal, soy flour, laver aquatic plant, kelp, oat flour, yeast, cod fish meal, fish liver, chlorophyll, vitamin A 12500 IU Kg-1, vitamin D3 1,500 IU Kg-1, vitamin E 25 mg Kg-1, vitamin B1 10 mg Kg-1, vitamin B2 20 mg Kg-1, vitamin B6 10 mg Kg-1, vitamin B12 0.6 mg Kg-1, folic acid 10 mg Kg-1, vitamin D-Pantothenic acid 10 mg Kg-1, vitamin C 100 mg Kg-1, lnositol 600 mg Kg-1, biotin 75 mg Kg-1 and niacin 50 mg Kg-1.
Guaranteed: Minimum crude protein 46%, minimum crude fat 5%, maximum crude fiber 2 % and maximum moisture 8%.
Nickel exposure: Preliminary tests were performed to determination the lethal toxicity of nickel, as nickel sulphate (NiSO4• 7H2O), at 24 h and was found to be 80 ppm. Thereafter, two groups of Oreochromis niloticus of 211-233 g weight, were randomly distributed (16 fish per group) in four glass aquaria. Fish of group one were exposed to 16 ppm of nickel for thee weeks. The experimental animals of group two were untreated and used as control.
Physiological determinations: At the end of experimental period, blood samples were collected by caudal peduncle cutting. Sera were separated by centrifugation at 2000 rpm for 15 min. and used for measuring sodium, chloride, osmolality, glucose, cholesterol, total protein, albumin, amylase, lipase, alanine aminotransferase (ALT) and aspartate aminotransferase (AST). All of these parameters were analyzed by using BM/Hitachi System, Automatic Analyzer.
Histological examination: Gills from each group were preserved in 10% buffered formalin. Tissues were processed, sectioned at four Fm and stained with hematoxylin and eosin, using standard histological techniques.
Statistical analysis: Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) version 12.0. Data were expressed by means±SD. Differences between means were evaluated by one-way analysis of variance (ANOVA). Statistical significance of the differences between means was assessed by Student's t-test. p≤ 0.05 was considered significance.
RESULTS AND DISCUSSION
In comparison with control data, the levels of serum sodium, chloride and osmolality were significantly diminished in the fish administrated with sublethal concentration of nickel. Serum glucose, cholesterol, total protein, albumin and the activities of amylase, lipase, ALT and AST were significantly elevated in nickel-exposed group in relation to control group, Table 1. Histopathological changes in epithelia were observed in gills of Oreochromis niloticus exposed to nickel (Fig. 3-7) in respect to normal structure of control group (Fig. 1-2). Hyperplasia, an increased proliferation of cells and hypertrophy were observed in animals exposed to nickel, shortening of secondary lamellae and occasionally resulted in fusion of adjacent lamellae and even of adjacent filaments.
The substantial surface area of the gills in fish serves as an interface between the environment and blood, notably for the continuous diffusion of oxygen and the maintenance of acid-base and ion balance (Randall et al., 1996; Claiborne et al., 2002).
| Table 1: | Influences of nickel administration on serum sodium, chloride, osmolality, glucose, cholesterol, total protein, albumin, amylase, lipase, ALT and AST levels in the fish, Oreochromis niloticus. n=8. |
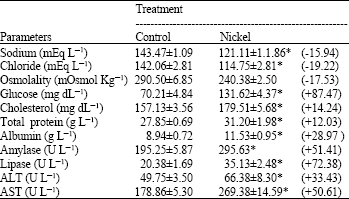 | |
| * Statistically significance at p≤ 0.05; Student's t-test + or – indicates an increase or a decrease percentage | |
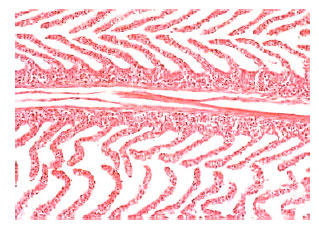 | |
| Fig. 1: | Normal primary lamellae of Oreochromis niloticus gill arch with a central hyaline cartilage, secondary lamellae perpendicular to the primary lamellae with abundant vascular spaces and a multi-layered interlamellar epithelium. Original magnification X100. |
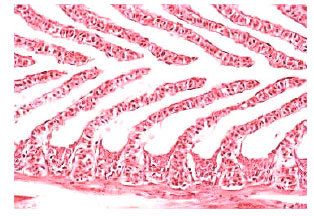 | |
| Fig. 2: | Secondary lamellae perpendicular to the primary lamellae and a multi-layered interlamellar epithelium in the normal gill of Oreochromis niloticus. Original magnification X400 |
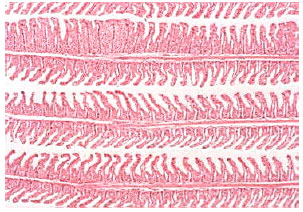 | |
| Fig. 3: | Gill lamellar epithelial hyperplasia, hypertrophy, shortening of secondary lamellae and with some lamellar fusion in nickel-exposed Oreochromis niloticus. Original magnification X40 |
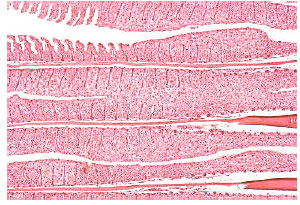 | |
| Fig. 4: | Severe gill lamellar epithelial hyperplasia, hypertrophy, shortening of secondary lamellae and with most lamellar fusion and adjacent filaments in nickel-treated Oreochromis niloticus. Original magnification X40 |
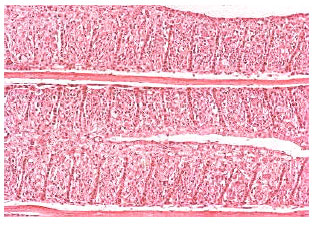 | |
| Fig. 5: | Severe gill lamellar epithelial hyperplasia, hypertrophy, shortening of secondary lamellae, complete lamellar fusion and most adjacent filaments in Oreochromis niloticus intoxicated with nickel. Original magnification X100 |
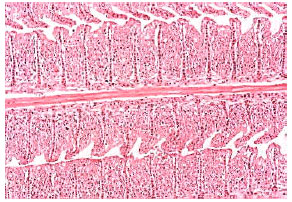 | |
| Fig. 6: | Severe gill lamellar epithelial hyperplasia, hypertrophy, shortening of secondary lamellae and with complete lamellar fusion in nickel-poisoned Oreochromis niloticus. Original magnification X200 |
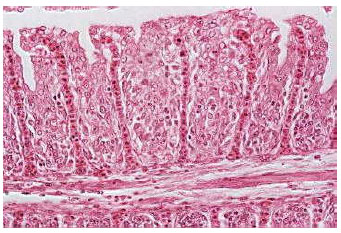 | |
| Fig. 7: | Severe gill lamellar epithelial hyperplasia, hypertrophy, shortening of secondary lamellae and with complete lamellar fusion in Oreochromis niloticus subjected to nickel. Original magnification X400 |
Fish that inhabit polluted environments are particularly susceptible to contaminants (soluble and suspended) that can damage gill structure and physiology (Lichtenfels et al., 1996; Randall et al., 1996). Metals and relatively low-molecular-weight organic compounds are readily absorbed across the gill (Randall et al., 1996). These contaminants cause deleterious changes in cellular structures, including the epithelium and pillar cells (Laurent and Perry, 1991; Randall et al., 1996; Van Veld et al., 1997). The present study showed that the gills of Oreochromis niloticus exposed to nickel during three weeks presented a higher occurrence of histopathological lesions such as hypertrophy, hyperplasia, shortening of secondary lamellae and fusion of adjacent lamellae and even of adjacent filaments. Similar of these changes in gill epithelia of Oreochromis niloticus were also structurally and ultrastructurally observed by Nath and Kumar (1989) and Pane et al. (2004) in the tropical freshwater perch, Colsia fasciatus and rainbow trout, Oncorhynchus mykiss, subjected to nickel; Ghazaly et al. (1994), Karan et al. (1998) and Mazon et al. (2002) in Cyprinion mhalensis, Cyprinus carpio and Prochilodus scrofa exposed to copper; Hemalatha and Banerjee (1997) in the air-breathing catfish, Hetropneustes fossilis intoxicated with zinc; De Boeck et al. (2001) in the spiny dogfish, Squalus acanthias poisoned with sliver; Gupta and Dua (2002) in mercury-administrated the air-breathing freshwater Chana punctatus; Thophon et al. (2003) and Rangsayatron et al. (2004) in Lates calcarifer and Puntius gonionotus treated with cadmium. In the study of Pane et al. (2004), nickel exposure resulted in marked ultrastructural damage to the respiratory epithelium of the gill in Oncorhynchus mykiss including swelling of the secondary lamellae evidenced by changes to both the lamellar region (increased secondary lamellar tissue volume) and to the secondary lamellae themselves (increased volume of tissue lying outside the pillar system). Additionally, decreased lamellar height and increased lamellar width indicated a reduction in lamellar surface area available for gas diffusion. Mallat (1985) stated that reflecting direct toxic action, lifting and hyperplasia of the lamellar epithelium could be interpreted as defense responses of the fish, as these alterations increase the distance across which waterborne irritants must diffuse to reach the blood stream. Epithelial lifting and hyperplasia of undifferentiated epithelial cells are known to be nonspecific alterations, which can be caused by a variety of unrelated insults such as those caused by heavy metals (Heath, 1987; Hinton et al., 1992; Randy et al., 1996). Exposure of fish to heavy metals may also result in variable degrees of ion regulatory disruption and plasma ion levels may be employed for quantifying toxic effects of metals intoxication (Mayer et al., 1992). McDonald and Wood (1993) stated that during exposure to waterborne heavy metals, the active uptake of ions from the water may be initially impaired, leading to ionic homeostasis disturbances. Pelgrom et al. (1995), Mazon et al. (2002) and Al-Attar (2006) reported that in Oreochromis mossambicus, Prochilodus lineatus and Cyprinion mhalensis exposed to copper, cadmium and mercury, blood sodium and chloride concentrations declined significantly. In freshwater fish, osmotic water influx and diffusive losses of ions such as sodium and chloride are compensated for by the excretion of large volumes of dilute urine and active uptake to replace ions lost by the gills (Evans et al., 1999). In constant contact with the water, the gill is a sensitive primary target for a variety of insults including heavy metals (Hinton et al., 1992). Given that the gills are the major sites of osmotic and ionic regulation in fish, any changes in gill morphology may result in perturbed osmotic and ionic status. Martinez et al. (2004) showed that the sodium decrease might reflect a decrease in the sodium influx rate. Na+/K+-ATPase is the prime mediator of ion transport across cellular membranes and plays a central role in whole body ion regulation in marine and estuarine animals. Na+/K+-ATPase has been reviewed and assessed as a potentially useful indicator of pollution stress in aquatic animals (Torreblanca et al., 1989). The mechanism of osmoregulatory disruption by metals normally involves inhibition of Na+/K+-ATPase enzymes in gills and perhaps in the gut as well. Therefore, lead might be inhibiting gill Na+/K+-ATPase on Prochilodus lineatus, what may cause a disruption on sodium hyperregulation. Ahern and Morris (1998) reported that the heavy metal, lead is a potent inhibitor of Na+/K+-ATPase activity, which was clearly demonstrated by a 50% reduction of Na+/K+-ATPase activity of the freshwater crayfish Cherax destructor incubated in vitro with 120 ppm lead. Moreover, Monterio et al. (2005) reported that gill copper accumulation in Oreochromis niloticus increased linearly with exposure time and concentration, whereas gill Na+/K+-ATPase activity was maximally inhibited after three days of exposure and showed a significant negative correlation with copper tissue levels. Plasma chloride values decreased with time of exposure. Plasma sodium and osmolality decreased with exposure time at the highest copper concentration tested.
Exposure of nickel induced a significant rise in the level of blood glucose in the experimental animal, Oreochromis niloticus. Several reports showed that the levels of blood glucose were significantly increased in the fishes treated with nickel, lead, copper, cadmium and mercury (Nath and Kumar, 1988; James et al., 1992; Jha and Jha, 1994; Van Vuren, 1994; Al-Attar, 2005; 2006). Several heavy metals have been reported to stimulate interrenal activity and plasma corticosteroid and glucose levels in fish (Pratap and Wendelaar Bonga, 1990). Hypersecretion of adrenalin and cortisol are considered primary stress responses. These effects trigger a broad suite of biochemical and physiological alterations called secondary stress responses. Metabolic effects include hyperglycaemia, depletion of tissue glycogen reserves, catabolism of muscle protein and altered blood levels of protein, cholesterol and free fatty acids (Thomas, 1990; Jobling, 1994; Wendelaar Bonga, 1997). Chaudhry (1984) stated that the muscle glycogenolysis accompanied by blood hyperlacticaemia is probably the result of some direct action of nickel on the fish, Colisa fasciatus and the sever stress conditions caused by nickel induced pathological changes in the gills and blood may also be responsible for these metabolic changes. Additionally, the present observations of hypercholesterolaemia, hyperproteinaemia and hyperalbuminaemia in the fish, Oreochromis niloticus after nickel treatment is possibly due to liver injury. Nickel-treated rats showed a significant increase in serum low-density lipoprotein-cholesterol, total cholesterol, triglycerides and a significant decrease in serum high-density lipoprotein-cholesterol. In the liver, nickel sulphate caused a loss of normal architecture, fatty changes, extensive vacuolization in hepatocytes, eccentric nuclei and Kupffer cell hypertrophy (Das et al., 2006). However, several studies showed that blood cholesterol and/or total protein were elevated in animals intoxicated with different metals (Jha and Jha, 1994; Gopal et al., 1997; Das and Dasgupta, 2002; Al-Attar, 2006; 2005). Determinations of serum lipase (triacylglycerol acylhydrolase; EC 3.1.1.3) and amylase (1,4-α-D-glucan glucanohydrolase; EC 3.2.1.1) activities are important in the diagnosis of acute pancreatitis (Lott et al., 1986; Lott and Lu, 1991; Yang et al., 2005). Yang et al. (2005) demonstrated that the changes of enzyme activity, hyperamylasemia and hyperlipasemia, might be caused by cancer-destructed excretory function, inflammation and gallstone, which lead excretion of enzymes into blood. The present hyperamylasemia and hyperlipasemia indicates pancreatic acinar cells injury in Oreochromis niloticus intoxicated with nickel. Several studies demonstrated that exposure to various stressors caused pancreatic damage, hyperamylasemia and hyperlipasemia (Prinz, 1991; Takacs et al., 1996; Cosen-Binker et al., 2003; Alhan et al., 2004; Zhang et al., 2005; Alsfasser et al., 2006; Cosen-Binker et al., 2006; Yan et al., 2006). ALT and AST are the most sensitive tests for diagnosis of liver diseases. The extend of hepatic damage is assessed by increased serum level of cytoplasmic enzymes, ALT and AST (Kew, 2000). Ptashynski et al. (2002) observed several histological alterations in the liver of lake whitefish, Coregonus clupeaformis, exposed to nickel. These alterations including focal necrosis and altered bile ducts. The present study revealed a significant increase in the activity of ALT and AST after three weeks of nickel exposure, indicating considerable hepatocellular damage. Similar observations and suggestions were stated by many experimental investigations on animals treated with heavy metals (Singh and Reddy, 1990; Karan et al., 1998; Bersenyi et al., 2003; 2004; Kim and Kang, 2004; Al-Attar, 2005).
From the current investigation, it can be stated that nickel produces severe toxic effects on the respiratory system, osmoregulation, liver and pancreas functions in the experimental animal, Oreochromis niloticus. Moreover, these physiological and histopathological changes indicate that the fish, Oreochromis niloticus is appropriate species to act as a biological indicator of water pollution level.
REFERENCES
- Ahern, M.D. and S. Morris, 1998. Accumulation of lead and its effects on Na balance in the freshwater crayfish Cherax destructor. J. Exp. Zool., 281: 270-279.
Direct Link - Al-Attar, A.M., 2005. Biochemical effects of short-term cadmium exposure on the freshwater fish, Oreochromis niloticus. J. Biol. Sci., 5: 260-265.
CrossRefDirect Link - Alhan, E., N.I. Kalyoncu, C. Ercin and B.V. Kurai, 2004. Effects of the celecoxib on the acute necrotizing pancreatitis in rats. Inflammation, 28: 303-309.
Direct Link - Alsfasser, G., A.L. Warshaw, S.P. Thayer, B. Antoniu and M. Laposata et al., 2006. Decreased inflammation and improved survival with recombinant human activated protein C treatment in experimental acute pancreatitis. Arch. Surg., 141: 670-677.
Direct Link - Bersenyi, A., S.G. Fekete, Z. Szocs and E. Berta, 2003. Effect of ingested heavy metals (Cd, Pb and Hg) on haematology and serum biochemistry in rabbits. Acta Veterinaria Hungarica, 51: 297-304.
CrossRefPubMedDirect Link - Bersenyi, A., S.G. Fekete, M. Szilagyi, E. Berta, L. Zoldag and R. Glavits, 2004. Effects of nickel supply on the fattening performance and several biochemical parameters of broiler chickens and rabbits. Acta Vet. Hung., 52: 185-197.
Direct Link - Brix, K.V., J. Keithly, D.K. DeForest and J. Laughlin, 2004. Acute and chronic toxicity of nickel to rainbow trout (Oncorhynchus mykiss). Environ. Toxicol. Chem., 23: 2221-2228.
Direct Link - Cosen-Binker, L.I., M.G. Binker, G. Negri and O. Tiscornia, 2003. Acute pancreatitis possible initial triggering mechanism and prophylaxis. Pancreatology, 3: 445-456.
Direct Link - Cosen-Binker, L.I., M.G. Binker, R. Cosen, G. Negri and O. Tiscornia, 2006. Relaxin prevents the development of severe acute pancreatitis. World J. Gastroenterol., 14: 1558-1568.
Direct Link - Damjanov, I., F.W. Sunderman, J.M. Mitchell and P.R. Allpass, 1978. Induction of testicular sarcoma in Fischer rats by intratesticular injection of nickel subsulfide. Cancer Res., 38: 268-276.
PubMed - Das, K.K. and S. Dasgupta, 2002. Effect of nickel sulfate on testicular steroidogenesis in rats during protein restriction. Environ. Health Perspect., 110: 923-926.
PubMed - Das, K.K., A.D. Gupta, S.A. Dhundasi, A.M. Pati, S.N. Das and J.G. Ambekar, 2006. Effect of L-ascorbic acid on nickel-induced alterations in serum lipid profiles and liver histopathology in rats. J. Basic Clin. Physiol. Pharmacol., 17: 29-44.
Direct Link - Doreswamy, K., B. Shrilatha, T. Rajeshkumar and Muralidhara, 2004. Nickel-induced oxidative stress in testis of mice: Evidence of DNA damage and genotoxic effects. J. Androl., 25: 996-1003.
PubMed - Gupta, N. and A. Dua, 2002. Mercury induced architectural alterations in the gill surface of a fresh water fish, Channa punctatus. J. Environ. Biol., 23: 383-386.
Direct Link - Gupta, A.D., A.M. Patil, J.G. Ambekar, S.N. Das, S.A. Dhundasi and K.K. Das, 2006. L-ascorbic acid protects the antioxidant defense system in nickel-exposed albino rat lung tissue. J. Basic Clin. Physiol. Pharmacol., 17: 87-100.
Direct Link - Hinton, D.E., P.C. Baumann, G.R. Gardner, W.E. Hawkins and J.D. Hendricks et al., 1992. Histopathological Biomarkers. In: Biomarkers: Biochemical, Physiological and Histological Markers of Anthropogenic Stress, Huggett, R.J., R.A. Kimerle, P.M. Mehrle-Jr. and H.L. Bergman (Eds.). Lewis Publishers, Boca Raton, Fl.
- Hoang, T.C., J.R. Tomasso and S.J. Klaine, 2004. Influence of water quality and age on nickel toxicity to fathead minnows (Pimephales promelas). Environ. Toxicol. Chem., 23: 86-92.
Direct Link - Kang, J., Y. Zhang, J. Chen, H. Chen, C. Lin, Q. Wang and Y. Ou, 2003. Nickel-induced histone hypoacetylation: the role of reactive oxygen species. Toxicol. Sci., 74: 279-286.
Direct Link - Karan, V., S. Vitorovic, V. Tutundzic and V. Poleksic, 1998. Functional enzyme activity and gill histology of carp after copper sulfate exposure and recovery. Ecotoxicol. Environ. Safety, 40: 49-55.
Direct Link - Kaur, P. and H.M. Dani, 2003. Carcinogenicity of nickel is the result of its binding to RNA and not to DNA. J. Environ. Pathol. Toxicol. Oncol., 22: 29-39.
Direct Link - Kim, S.G. and J.C. Kang, 2004. Effect of dietary copper exposure on accumulation, growth and hematological parameters of the juvenile rockfish, Sebastes schlegeli. Mar. Environ. Res., 58: 65-82.
Direct Link - Laurent, P. and S.F. Perry, 1991. Environmental effects on fish gill morphology. Physiol. Zool., 64: 4-25.
Direct Link - Lichtenfels, A.J.F.C., G. Lorenzi-Filho, E.T. Guimaraes, M. Macchione and P.H.N. Saldiva, 2006. Effects of water pollution on the gill apparatus of fish. J. Comp. Pathol., 115: 47-60.
Direct Link - Mallatt, J., 1985. Fish gill structural changes induced by toxicants and other irritants: A statistical review. Can. J. Fish. Aquat. Sci., 42: 630-648.
CrossRefDirect Link - Martinez, C.B.R., M.Y. Nagae, C.T.B.V. Zaia and D.A.M. Zaia, 2004. Acute morphological and physiological effects of lead in the neotropical fish Prochilodus lineatus. Braz. J. Biol., 64: 797-807.
CrossRefDirect Link - Mayer, F.L., D.J. Versteeg, M.J. Mckee, L.C. Folmar, R.L. Graney, D.C. Mccume and B.A. Rattner, 1992. Physiological and Non-specific Biomarkers. In: Biomarkers: Biochemical, Physiological and Histological Markers of Anthropogenic Stress, Huggett, R.J., R.A. Kimerle, P.M. Mehrle-Jr. and H.L. Bergman (Eds.). Lewis Publishers, Boca Raton, Fl.
- Mazon, A.F., E.A.S. Monteiro, G.H.D. Pinheiro and M.N. Fernandes, 2002. Hematological and physiological changes induced by short-term exposure to copper in the freshwater fish, Prochilodus scrofa. Braz. J. Biol., 62: 621-631.
CrossRefDirect Link - Monteiro, S.M., J.M. Mancera, A. Fontinhas-Fernandes and M. Sousa, 2005. Copper induced alterations of biochemical parameters in the gill and plasma of Oreochromis niloticus. Compa. Biochem. Physiol. Part C: Toxicol. Pharmacol., 141: 375-383.
CrossRefDirect Link - Pane, E.F., C. Smith, J.C. McGeer and C.M. Wood, 2003. Mechanisms of acute and chronic waterborne nickel toxicity in the freshwater cladoceran, Daphnia magna. Environ. Sci. Technol., 37: 4382-4389.
Direct Link - Pratap, H.B. and S.E.W. Bonga, 1990. Effects of water-borne cadmium on plasma cortisol and glucose in the cichlid fish Oreochromis mossambicus. Comp. Biochem. Physiol. Part C: Comp. Pharmacol., 95: 313-317.
CrossRefDirect Link - Ptashynski, M.D., R.M. Pedlar, R.E. Evan, C.L. Baron and J.F. Klaverkamp, 2002. Toxicology of dietary nickel in lake whitefish (Coregonus clupeaformis). Aquat. Toxicol., 58: 229-247.
CrossRef - Randi, A.S., J.M. Monserrat, E.M. Rodriguez and L.A. Romano, 1996. Histopathological effects of cadmium on the gills of the freshwater fish, Macropsobrycon uruguayanae Eigenmann (Pisces, Atherinidae). J. Fish. Dis., 19: 311-322.
CrossRefDirect Link - Rangsayatron, N., P. Pokethitiyook, E.S. Upatham, G.R. Lanza and S. Singhakaew, 2004. Ultrastructural changes in various organs of the fish Puntius gonionotus fed cadmium-enriched cyanobacteria. Environ. Toxicol., 19: 585-593.
Direct Link - Singh, H.S. and T.V. Reddy, 1990. Effect of copper sulfate on hematology, blood chemistry and hepato-somatic index of an Indian catfish, Heteropneustes fossilis (Bloch) and its recovery. Ecotoxicol. Environ. Saf., 20: 30-35.
Direct Link - Stentiford, G.D., M. Longshaw, B.P. Lyons, G. Jones, M. Green and S.W. Feist, 2003. Histopathological biomarkers in estuarine fish species for the assessment of biological effects of contaminants. Mar. Environ. Res., 55: 137-159.
CrossRefPubMedDirect Link - Thophon, S., M. Kruatrachue, E.S. Upatham, P. Pokethitiyook, S. Sahaphong and S. Jaritkhuan, 2003. Histopathological alterations of white seabass, Lates calcarifer, in acute and subchronic cadmium exposure. Environ. Pollut., 121: 307-320.
CrossRefDirect Link - Van Vuren, J.H., M. van der Merwe and H.H. du Preeze, 1994. The effect of copper on the blood chemistry of Clarias gariepinus (Clariidae). Ecotoxicol. Environ. Safety, 29: 187-199.
PubMed - Bonga, S.E.W., 1997. The stress response in fish. Physiol. Rev., 77: 591-625.
CrossRefPubMedDirect Link - Yan, O., X. Yao, L.C. Dai, G.L. Zhang, J.L. Ping, J.F. He and C.F. Han, 2006. Effect of early administration of exogenous basic fibroblast growth factor on acute edematous pancreatitis in rats. World J. Gastroenterol., 21: 3060-3064.
Direct Link - Yang, R.W., Z.X. Shao, Y.Y. Chen, Z. Yin and W.J. Wang, 2005. Lipase and pancreatic amylase. activities in diagnosis of acute pancreatitis in patients with hyperamylasemia. Hepatobilliary Pancreat. Dis. Int., 4: 600-603.
Direct Link








