Research Article
Proteolytic Activity of Bacillus cereus Under in vitro Condition
Department of Biotechnology, K.S. Rangasamy College of Technology, Tiruchengode- 637 209, Namakkal District, Tamilnadu, India
Bacillus genus contamination in foods leads to food poisoning and food intoxication. Bacillus cereus gastroenteritis an exoentero toxin released during lysis of B. cereus in the intestinal tract (Omafuvbe et al., 2002). Degradation of proteins into amino acids or peptides named as proteolysis. Heterogeneous group of actively photolytic bacteria produces extra cellular proteinases. So, it is termed because the enzymes diffuse outside the cell and cause proteolysis. All bacteria have proteinases inside the cell, but only a limited number of kinds have extra cellular proteinases. In Bacillus genus, most of the species have the ability to proteolysis but the amount of proteolysis is different among species (Biedrzycka et al., 2003).
Protease is one of the most important classes of industrial enzymes and playing important role in biotechnology. It has been explored in oleochemical industry especially for fermentation (Hittu and Punj, 1996). Proteases have been used extensively in food, dairy, beverages and detergent industries (Aaku et al., 2005). In this present study, water, egg granules and juice packs were collected for the analysis. From which, attempts were made to isolate microorganisms and studied proteolysis activity under lab condition.
MATERIALS AND METHODS
The study was conducted at School of Biotechnology, K.S. Rangasamy College of Technology, Tiruchengode, Namakkal District of Tamilnadu, India, for a period of two years (2005-2006). There were three test samples such as water, egg granules and flavour juices used for the present study. Quantitative enumeration of microorganisms in test samples was done by serial dilution by agar plate method. Water and juice samples were diluted into different concentrations. In contrasts, for egg samples 10 g of the samples were dissolved in 100 mL of sterile distilled water to achieve 10-1 dilution. Ten milliliter of the 10-3 dilutions from all samples were taken and 90 mL of the standard plate count agar was mixed and, this mixture was kept in water bath for 30 min at 80°C. This was poured into five sterile petriplates. Finally these plates were allowed to solidify and were incubated at 37°C for 24 h.
In order to isolate Bacilli species from the test samples, 10-3 diluted samples were spread plated on SMCA medium and plates were incubated at 37°C for 24-48 h. After incubation period, mannitol fermenting and non-mannitol fermenting colonies were picked for confirmation of Bacillus species. Isolates obtained from water samples were designated as WB-1, WB-2. Similarly, isolates obtained from egg granules were designated as EB-1 and EB-2 and isolates from juices were named as JMB (Mango), JLB (Lemon) and JGB (Grape) subsequently.
To characterize the isolates, over night culture were prepared and they were subjected to analyse biochemical tests such as catalase, Lecithovitellin/Lecithiase production, citrate utilization, VP reaction, nitrate reduction, indole production, starch hydrolysis, amylase production, urease activity, catalase activity and sugar fermentation (glucose, fructose, maltose, sucrose, lactose, cellobiose, mannitol, xylose and arabinose) and morphological tests such as gram staining, acid fast staining and motility.
The isolates were screamed for proteolytic activity by using egg albumin agar and skimmed milk casein agar media. The inoculated plates were incubated for a period of 48-72 h at 37°C. The activity of proteolysis was observed by formation halo zone around the colonies, which indicated proteins were hydrolyzed by the bacteria.
The results on the enumeration of Bacilli showed that the number of colonies were found to be more in water samples followed by egg granules and it was least in flavour juice samples (Table 1). The average of microbial load (Bacilli colonies) in water samples was 521.4x10-3 cfu mL-1.
| Table 1: | Enumeration of heat resistant Bacilli species in different test samples |
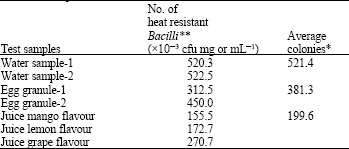 | |
| * x10-3 cfu mg or mL-1, **Average of three replicates, Water and juice samples -x10-3 cfu mL-1, Egg granule samples -x10-3 cfu mg-1 | |
Similarly, the average of microbial load in egg granules samples was 381.3x10-3 cfu mg-1 and it was 199.6x10-3 cfu mL-1 flavour juices. Among the three samples of flavour juice tested, the microbial load was found to be higher in grape flavour (270.7x10-3 cfu mL-1) followed by lemon (172.7 x10-3 cfu mL-1) and it was very least in mango flavour sample (155.5x10-3 cfu mL-1). Similar observations were made by Dgrak et al. (1996) and who examined 21 cheese samples. The results revealed that the microbiological quality and fatty acids composition of fresh white cheeses contained an average counts of Coliform bacteria, Staphylococcus aureus, Bacillus, Psychrophilic bacteria, yeast/moulds and heterofermentative lactic acid bacteria was reported.
The results on morphological tests indicated that all the isolates were gram positive and motile in nature (Table 2). They were well spore forming and grew well under anaerobic condition. All the isolates were found to be positive in catalase, nitrate reduction, citrate reduction, starch hydrolysis, amylase production and Vogesproskaur reaction tests and negative for indole production by most of the isolates. Based on the above tests, all the isolates are belonging to Bacillus cereus. A similar observation was reported by HittuMatta and Punj (1999) and Lindsay et al. (1999). Moges and Ashenafi (2000) were examined one hundred twenty five samples of juices for the incidence and level of contamination of Bacillus species. They were reported that contamination was due to Bacillus cereus.
The activity of proteolysis was measured by the formation of halo zones around the colony in the medium. The size of halo zone around colonies indicated that it was showed higher in albumin agar medium inoculated with WB-2 (32.3 mm) showed followed by WB-1 (30.5) and least by JGB (22.3 mm) isolate. Similarly, the size of halo zone around colonies in skim milk agar medium showed higher by WB-2 (35.5 mm) followed by WB-1 (32.5) and EB-2 (32.3) and least by JGB (25.7) isolate (Table 3).
| Table 2: | Characterization of isolates obtained from test samples |
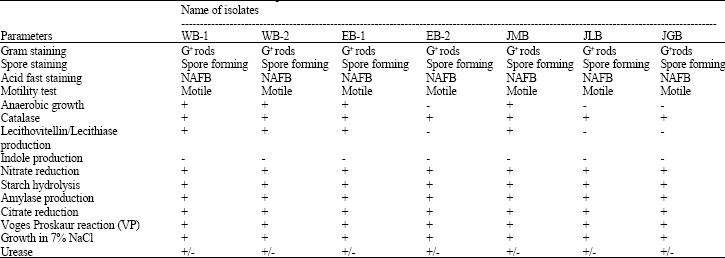 | |
| G+ rods: Gram positive rods. +: Positive , - : Negative, NAFB: Non-acid fast Bacilli, +/- : Variable | |
| Table 3: | Studies on the activity of proteolysis by the isolates |
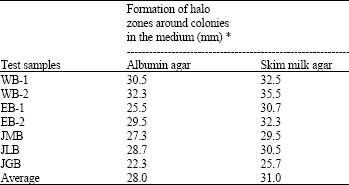 | |
| * Average of three replicates | |
Among the two different media such as albumin agar and skimmed milk agar tested, skimmed milk agar medium was found to be higher in term of the formation. It was found that the inhibition of proteolytic activity is a possible mechanism for enhanced xylanase activity reported by Balakrishnan et al. (1997) and Belima et al. (2002). In Bacillus genus, most of the species have the ability to proteolysis but the amount of proteolysis is different among species to species (Biedrzycka et al., 2003; Aaku et al., 2004). Some of the species of Bacillus have the ability to hydrolyze fats and other derivative compounds which results into rancidity in some food products (HittuMatta and Punj, 1999). The ability of Bacillus genus to hydrolyze proteins is accomplished due to the enzyme protease.
It may concluded that all the test samples were of poor hygienic quality. Similar attempt was made by Pirttijarvi et al. (1996) and Belima et al. (2002) who reported that the liquid packaging boards and blanks, such as those used to package of milk and other flavour juice products, were examined for microbial contaminants.
The authors are thankful to the Principal and the Management of K.S. Rangasamy College of Technology, Tiruchengode, Tamilnadu, India, for providing necessary facilities and constant encouragement to carry out this study.