Research Article
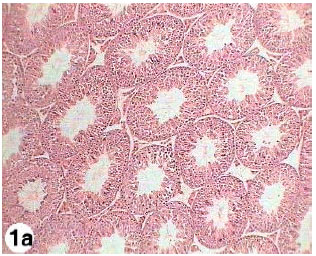
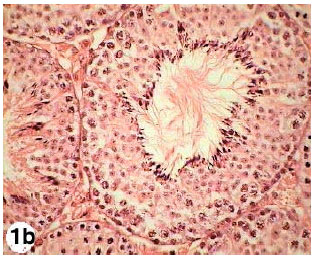
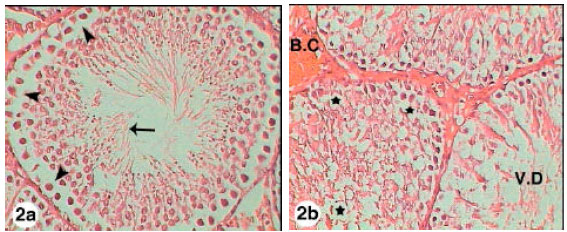
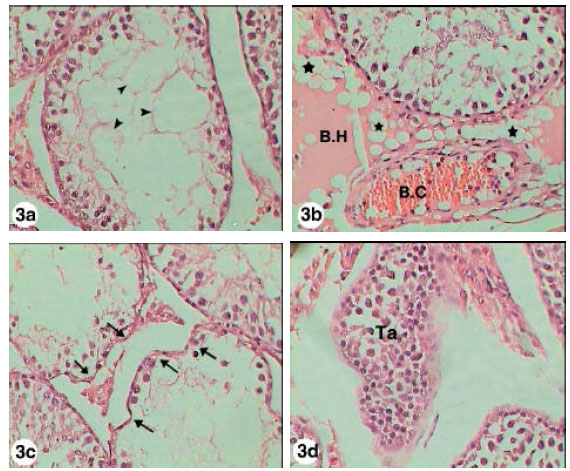
Testicular Histopathological Alterations in Rats Treated with Sumithion® NP 25/2.5 EC, Insecticide
Department of Sciences, Jeddah Teachers College, P.O. Box 15758, Jeddah, Saudi Arabia
Ameen S. Bin Bisher
Department of Sciences, Jeddah Teachers College, P.O. Box 15758, Jeddah, Saudi Arabia