Research Article
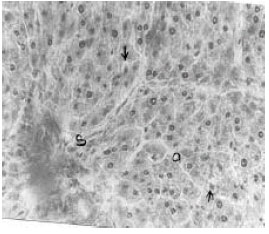
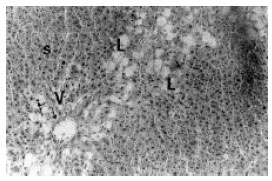
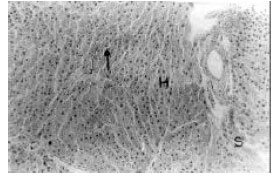
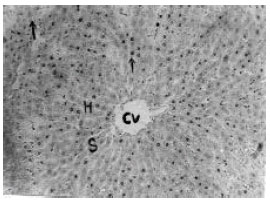
Hepatoprotective Effect of the Aqueous Root-Bark Extract of Ficus sycomorus (Linn) on Carbon Tetrachloride Induced Hepatotoxicity in Rats
Department of Veterinary Pharmacology and Physiology, Faculty of Veterinary Sciences, University ofMaiduguri, Nigeria
J. Prasad
Department of Veterinary Pharmacology and Physiology, Faculty of Veterinary Sciences, University ofMaiduguri, Nigeria
U. K. Sandabe
Department of Veterinary Pharmacology and Physiology, Faculty of Veterinary Sciences, University ofMaiduguri, Nigeria