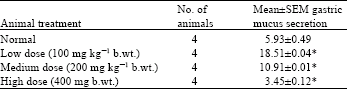Research Article
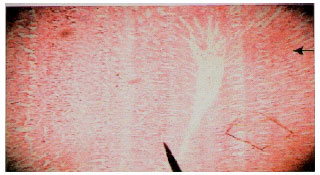
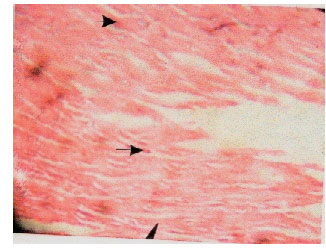
Methanolic Extract of Entandrophragma angolense Induces Gastric Mucus Cell Counts and Gastric Mucus Secretion
Department of Physiology, College of Medicine, University of lbadan, Ibadan, Nigeria
B. O. Omolaso
Department of Physiology, College of Medicine, University of lbadan, Ibadan, Nigeria
J.A. Ayo
Department of Physiology, College of Medicine, University of lbadan, Ibadan, Nigeria