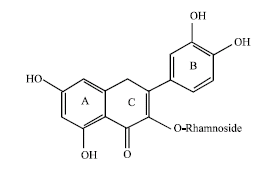
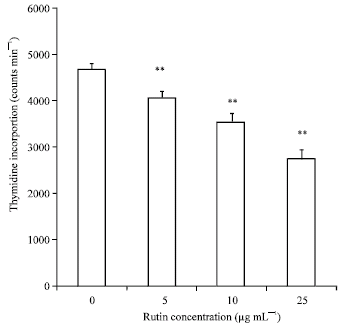
Research Article
Antiangiogenic Effect of Rutin and its Regulatory Effect on the Production of VEGF, IL-1β and TNF-α in Tumor Associated Macrophages
Department of Immunology, Amala Cancer Research Centre, Amala Nagar, Thrissur, Kerala State 680 555, India
Girija Kuttan
Department of Immunology, Amala Cancer Research Centre, Amala Nagar, Thrissur, Kerala State 680 555, India