Research Article
Root Apical Meristem Characteristics of Two Canola (Brassica napus L.) In Response to Salt Stress
Department of Biology, Faculty of Science, Islamic Azad University of Mashad, Iran
Salt stress is one of the most serious problems in agriculture in arid and semi-arid areas. A high concentration of NaCl greatly reduces growth of both the shoot and the root (Greenway and Munns, 1980; Cheesman, 1988). Unfortunately, the cellular mechanisms of salt injury in roots are scarcely known. Among the root cells, meristematic cells are especially interesting, because mitotic activity and cell division are indispensable for root growth and because meristematic cells are considered to be one of the salt-sensitive cells (Huang and Stereninck, 1988). These cells have been studied under exposure to moderate salinity (El-banna and Attia, 1999; Huang and Stereninck, 1990), but not under severe salt stress. In the present study, we study cell characteristics of the root apical meristem of two canola cultivars (salt-sensitive and salt-tolerant cv.) under the effect of excessive saline conditions.
The study was conducted at Faculty of Science, Mashad Azad University (2005). The seeds of canola were obtained from Agricultural Research Center of Khorasan, Iran. Seeds of Okapi (salt-tolerant cv.) and Symbol (salt-sensitive cv.) were germinated for 3 days in the dark at 25°C, on filter paper soaked with distilled water (control) or salt solution (3, 6, 9 and 12 dS m-1). For paraffin sections, root samples were fixed overnight in 10% formaline, 5% acetic acid and 50% ethanol (FAA). Samples were dehydrated in a graded ethanol series then embedded in paraplast, then were sectioned at 8 intervals and stained with hematoxylin-eosine. The distance from the root cap boundary to the position of the 1st root hair was measured on three roots per cultivar per treatment. For semithin sections, root samples were fixed in a mixture (4°C , 45 mM) of glutaraldehyde buffered (pH 7.4) with 0.2 M sodium cacodylate followed by a solution (45-60 min) of 1% (w/v) osmium tetroxide buffered (pH 7.4) with 0.1 M veronal, then they were included in araldite. Longitudinal sections (0.5 in thickness) were cut in three different roots of each type of plant using ultramicrotome then they were stained with toluidine blue (1%, w/v) for 5 min followed by rinsing in distilled water. In each root under study, the measurements were carried out using a microscope equipped with an ocular micrometer. The size of the meristem area (micron along root length) and the mitotic index (percentage of dividing cells relative to the total number of cells at each site of measurement) were thus obtained by averaging the values measured in the three different roots.
In root apical meristem of angiosperms including canola, three zones are recognizable:
| • | Calyptrogen that makes root cap and epidermis in dicotyledons. |
| • | Quiescent center that plays a supporting and regenerating role. |
| • | Active meristematic zone (Fig. 1). |
Root apical meristem of control plants had more homogenous chromophily in comparison with the treated samples. These features is probably a symptom of more active mitotic divisions in the meristem of control plants (Fig. 2 and 3).
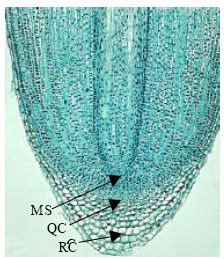 | |
| Fig. 1: | Longitudinal section of root apical meristem of angiosperm plant: three zones are recognizable: Active Meristematic zone (MS), Quiscent Center (QC) and Root Cap (RC) |
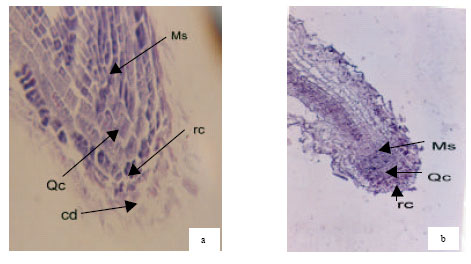 | |
| Fig. 2: | Longitudinal section of apical root of canola (Okapi cv.). Hematoxylin-eosine staining (a) control plant and (b) treated plant. Ms-active meristematic zone. Qc: Quiscent center, rc: root cap, cd: seperating cells from root cap |
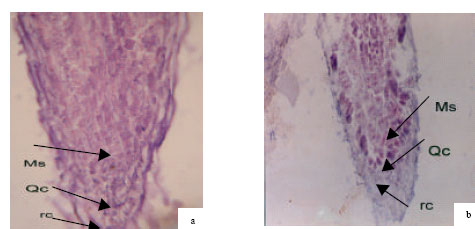 | |
| Fig. 3: | Longitudinal section of apical root of canola (Symbol cv.). Hematoxylin-eosine staining (a) control plant and (b) treated plant. MS-active meristematic zone. Qc: Quiscent center and rc: root cap |
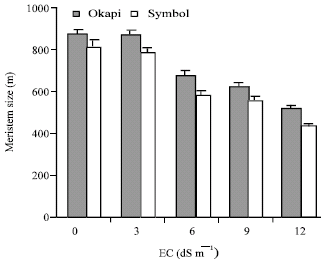 | |
| Fig. 4: | The meristem size in seedlings of canola cv. Okapi and symbol in the presence of NaCl different concentrations .the vertical bars represent +SE |
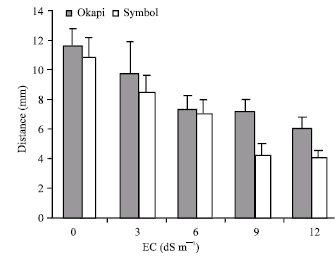 | |
| Fig. 5: | The distance from the root cap to the first root hair in seedlings of canola cv. okapi and symbol in presence of NaCl different concentrations |
The meristem size of both cultivars was markedly reduced by increasing NaCl concentration especially in the salt-sensitive cultivar (Fig. 4). The meristem size of Okapi was greater than Symbol in the control and all NaCl concentrations. The size of the root meristem was already known to be reduced as a consequence of treatment by herbicides (Elmore and Bayer, 1992) or a toxic metal (Ryan et al., 1993).
The distance of first root hair from the root cap boundary showed a marked decline in both cultivars with increasing NaCl concentration. NaCl induced reduction was more pronounced in Symbol than in Okapi (Fig. 5). This distance between the 1st root hair from the root cap boundary was reduced to 15.2 and 20.9% in comparison with the control at 3 dS m-1 treatment in Okapi and Symbol, respectively. The corresponding figure at 6 dS m-1 was 36.9 and 33.9%, respectively declining to 38.2 and 60.5% at 9 dS m-1. There was a further reduction to 47.8 and 62.7% in comparison with the control at 12 dS m-1.
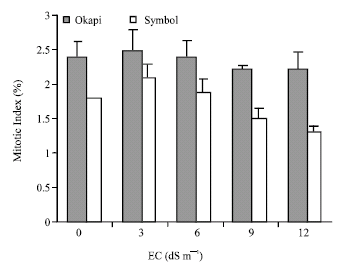 | |
| Fig. 6: | Mitotic index of the root meristem cells in seedlings of canola cv. okapi and symbol in presence of NaCl different concentrations the vertical bars represent +SE |
The mitotic index, which was smaller in the salt-sensitive than in the salt-tolerant cultivar was reduced in the two cultivars under salt stress (Fig. 6).
When grouping our present and previous data (Jbir et al., 2001a, b) the salt effects appear to be usually more pronounced in the roots of the salt-sensitive than the salt-tolerant canola cultivars. The mechanism of salt action is still not clear. NaCl effect might be indirect, via the disturbance of the distribution of essential other ions. In dicotyledone roots, transfer cells exhibiting a characteristic structure were observed to be present close to the vessels (Kramer et al., 1977) where they possibly involved in exclusion of sodium from the shoots. Increasing NaCl concentrations markedly reduced the size of root apical meristem in canola cultivars. The decreasing of the apical meristem size which was brought about by NaCl in both canola cultivars indicated that NaCl changed the balance that normally exists between cell division and the onset of elongation and differentiation Hwang and Chen (1995) showed that Kandelia candel grown in high salinities developed shorter roots than those grown in low salinities and suggested that the increased dimensions in the root apical region of K. candel is caused by an inhibition of cell division in the apical meristem. The reduction in meristem size at higher NaCl treatments reported here indicated that NaCl reduced the reproductive life span of cells in the meristem and leads to premature cell elongation. This phenomenon seems to be wide spread in the response to stress factors in roots.
For example, Ivanov (2006) showed that X-ray treatment of roots of corn caused premature cell elongation with shortening of meristem length. Davies et al. (1992) showed that zinc treatment reduced the length of apical meristem in the roots of Festuca rubra, the zinc-induced reduction was much more pronounced in a zinc sensitive cultivars (s59) than in a zinc tolerant cultivar (Merlin). Thomas (1992) showed that treatment with manganese reduced meristem length in roots of Epilobium hirsutum and Chamerion angustifolium and that the length of apical meristem of manganese treated root progressively increased in size. The precise mechanisms by which these stress factors reduce meristem length is not clear. It may be related to their effect in inhibiting cell division (Thomas, 1992) although Ivanov (2006) showed that the suppression of cell division by using a variety of anti-mitotic drugs did not affect the transition of cells to elongation. Ivanov (2006) suggested that in normal roots, the cessation of division and the beginning of elongation regulated by different mechanisms. There were marked differences between the cultivars in the position of the first root hair in relation to the root tip in the control (Ec = 0). This implies that the onset of cell differentiation (as measured by root hair formation) may have different positional controls in the cultivars. Powell et al. (1988) have shown that essentially similar pattern of change in the relative position of the most distal root hair and the most distal xylem element occurs in the root of Festuca rubra exposed to zinc. Increasing NaCl concentration resulted in the most distal root hair being formed progressively closer to the root tip in the cultivars. There was nearly 2.5-fold shortening of he distance to the most distal root hair over the range of NaCl concentration used (0-12 dS m-1) in Symbol. The lowest reduction (1.9-fold) occurred in Okapi. There was an indication that the cultivars under study were differentially affected by NaCl-induced reduction. Transition points for cell formation, cell transition to elongation and finally termination of growth leading to maturation are believed to operate within cell files (Ivanov, 2006). Rost and Baum (1988) demonstrated that these transition points were variable and depended on growth conditions. In summary, NaCl treatment can alter the meristem size, mitotic index and distance from the root tip to the first root hair. Contrasting cultivars were differently affected by NaCl in respect of these characters.