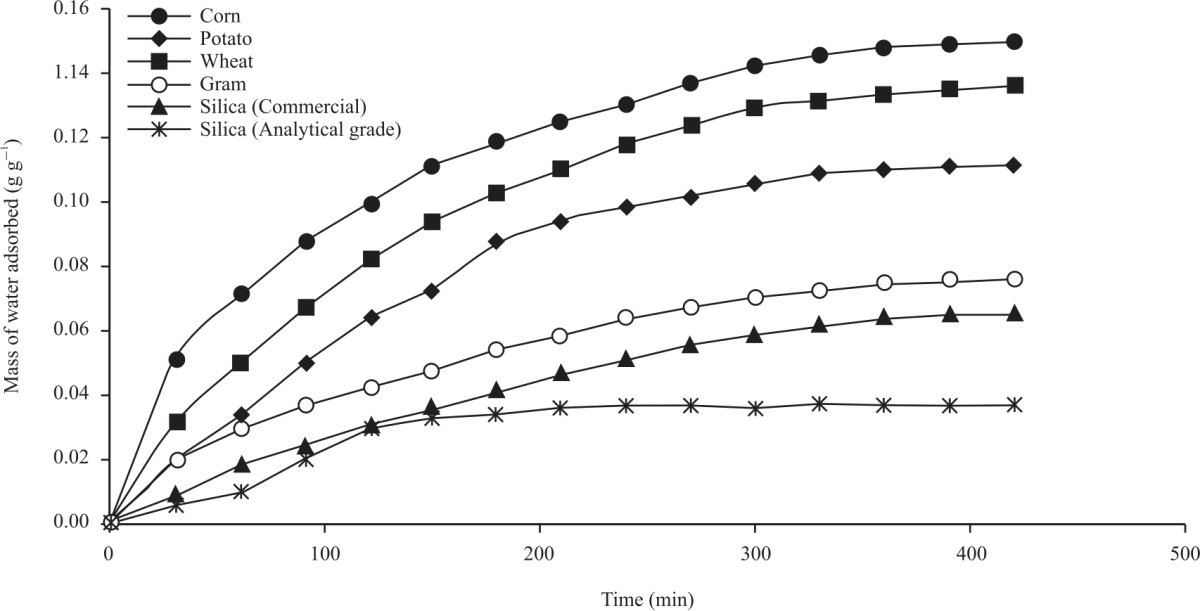
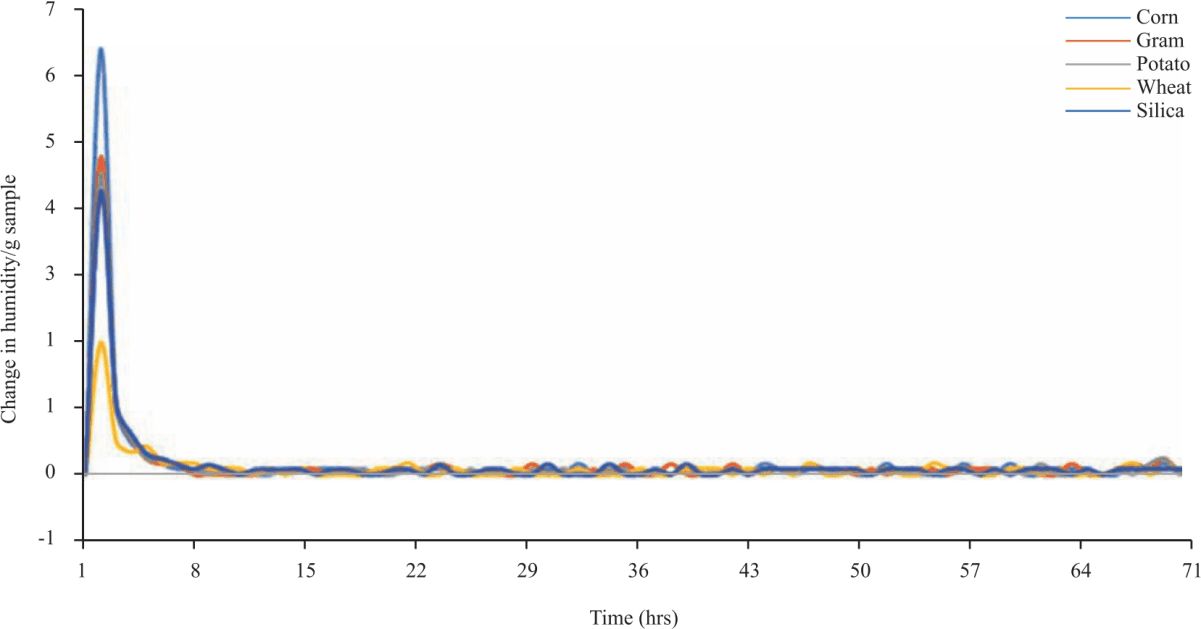
Research Article
Desiccant Properties Comparison of Natural Bio-polymers
Department of Science, Faculty of Engineering and Science, University of Agder, Norway
LiveDNA: 47.34985
Thilini Nisansala Rathnaweera
Department of Chemistry, Faculty of Science, University of Ruhuna, Matara, Sri Lanka
Kalana Dasunpriya Halanayake
Department of Chemistry, Faculty of Science, University of Ruhuna, Matara, Sri Lanka