Research Article
Physico-chemical, Fatty Acids and Amino Acids Composition of the Seeds of African Grapes (Lannea microcarpa) Fruits
Department of Chemistry, Sokoto State University, Sokoto, Sokoto State, Nigeria
LiveDNA: 234.23104
Wild fruits have helped to provide a steady supply of fruits during the dry season when cultivated fruits are scarce and expensive for low-income earners that traditionally have large family1. Millions of people in many developing countries do not have enough food to meet their daily requirements and most people are deficient in one or more micronutrients2.
In Nigeria, many rural communities depend on the wild resources including wild edible plants to meet their food needs in periods of food crisis, as well as for additional food supplements3. Fat is an essential part of the diet as it provides the body with energy in a concentrated form. Also, dietary fats provide essential fatty acids and fat-soluble vitamins. Unsaturated fatty acids especially Linoleic acid and linolenic acid are the essential fatty acids required in the diet4. These essential fatty acids have many functions in the body, for example, in the immune system and cell membranes, for the function of the brain and skin and may also reduce the risk of heart disease5. Among the food sources of essential fatty acids are fish, oils, kernels and nuts. The consumption of foods rich in monounsaturated fatty acids has been associated with health promoting effects, for example decreased levels of low density lipoprotein cholesterol and possibly increased high-density lipoprotein cholesterol4. Protein is another important nutrient in the diet. Inadequate supply of protein is considered to be responsible for malnutrition among people living in developing countries. Different beans, walnuts, peanuts and some seed kernels are the common sources of non-meat protein6.
African grapes (Lannea microcarpa) belong to the family Anacardiaceae and is one of the locally important tree species associated with crops in agroforestry parklands and also protected in home gardens in most part of the country. Its fruits are consumed fresh or used to make juice and fermented drink. Its bark is used for dyeing cotton fabrics in red-brown and in traditional medicine7. The seeds serve in vegetable oil production that is used in human and animals feeding and cosmetics8. The fruits of Lannea microcarpa are used for biodiesel production which physicochemical properties are similar to petro-diesel and that derived from Jatropha curcas9.
Information on the nutritional compositions of Lannea microcarpa fruits is very scanty and have received less attention, particularly in this part of the country (Northwest). Their contribution to local diets is less understood and quantified. Therefore, the study was carried out to determine the physicochemical and fatty acids composition of the seed oil as well as the amino acids content of the seed kernel with the aim of providing its potential use as nutrients supplement in the formulation of animal feeds and as an additional raw material in cosmetic industries.
Sampling and sample treatments: Fresh fruits of Lannea microcarpa were collected from Chimola district of Gwadabawa Local government Area, Sokoto state, Nigeria. The site was chosen because of the abundance of this plant. The fruits were collected from different branches of the selected trees, as described in the method of Ayaz et al.10. The sample was collected in black polythene bags and transported to laboratory. Prior to analyses, the sample was authenticated at the Herbarium section, Botany Unit, Department of Biological Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria. Representative sample was taken using alternate shovel method11. The sample was thoroughly washed with distilled water and then air dried. The peels and the pulp were removed manually using a sharp blade, while the kernels were removed manually using hammer, pulverized to fine powder and then used for the analyses12.
Physicochemical properties of the seed oil: The percentage oil yield, specific gravity, refractive index, viscosity, iodine value, acid value, saponification value and peroxide value were determined using the standard analytical methods described by AOAC13, while the acid value and percentage free fatty acids were determined using the method of Oderinde et al.14.
Fatty acids composition using GC/MS: The fatty acid analysis was carried out by injecting the clear supernatant of the fatty acid methyl esters (FAMEs) in to a splitless injector interfaced 5973 mass selective detector gas liquid chromatogram equipped with a detector. The fatty acids were observed as peaks whose retention times were measured by the spectrometer detector and compared with those of known standards of the Wiley library15.
Amino acids composition: The profile and the amount of total amino acids was determined by reverse phase HPLC, using the Pico-Tag system described by Trumbo et al.16. The sample was first defatted and hydrolyzed. For hydrolysis, 0.4 g of defatted sample placed in a flask and 15 mL of 6 M HCl added and the whole placed in an oven at 110°C for overnight. The sample are subsequently cooled to room temperature and transferred to a 50 mL volumetric flask and filled with Milli-Q water. Approximately, 1 mL of the diluted solution is homogenized and filtered through a filter of 0.45 μm. An aliquot of 10 μL of the solution is placed in reaction tube and dried for 15 min with the Picotag Workstation. The sample was then re-dissolved in 10 μL of re-drying solution (ethanol: water: triethylamine, 2:2:1 volume). It was dried again for 15 min and finally derivatized with 20 μL phenyl isothiocyanate reagent (ethanol:water:triethylamine:phenyl isothiocyanate, 7:1:1:1) for 20 min at room temperature. Excess reagent was removed with the aid of vacuum for 45 min in Pico-Tag Workstation. Derivatized samples were dissolved in 100 μL PicoTag Sample diluent solution (WAT088119). Analysis (identification and quantification) of amino acids performed using a Waters C18 column under the conditions described by Bidlingmeyer et al.17.
Briefly, 40 μL of aliquot was injected onto the column. Quantitation of amino acids was performed using a Waters C18 column (3.9×150 mm) with gradient conditions as described elsewhere. Derivatized amino acids were eluted from the column with increasing concentrations of acetonitrile. The eluate was monitored at 254 nm and the areas under the peaks were used to calculate the concentrations of the unknowns using a Pierce Standard H amino acid calibration mixture (Rockford, IL).
Data analysis: The data obtained was statistically analyzed using one-way analysis of variance (ANOVA) with SPSS version 16.0 statistical package and the results were reported as mean±standard deviation of the values.
The result of physicochemical properties and fatty acids composition of Lannea microcarpa seed oil is presented in Table 1 and 2, respectively, while the amino acids content of the seed kernel is presented in Table 3 and 4.
Physicochemical characteristics: The result of physicochemical properties of Lannea microcarpa seed oil is presented in Table 1. The oil contents was 48.23±2.43/100 g, which is lower than 64.90/100 g recorded for Lannea microcarpa seed oil of Burkina Faso18, this could be due to the genetic variations of plants as well as the climatic conditions. The value is higher than that of Adansonia digitata seed oil (29.33/100 g) reported by Warra et al.19. The result indicated that the Lannea microcarpa seed could be an additional raw material as a source of vegetable oil.
The specific gravity of the oil was found to be 0.95±0.10 g cm–3. The value recorded is comparable to 0.91 and 0.94 g cm–3 for seed oil of Lannea microcarpa and Adansonia digitata, respectively18,19. The value obtained shows that the oil is less dense than water.
| Table 1: | Physicochemical properties of Lannae microcarpa seed oil |
 | |
| The values are mean±standard deviation of three replicates | |
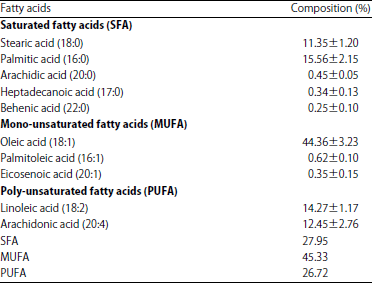
| Table 2: | Fatty acids composition of Lannea microcarpa seed oil |
 | |
| The values are mean±standard deviation of two replicates | |
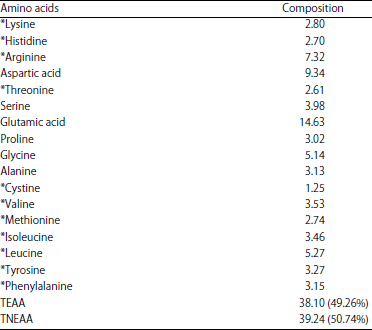
| Table 3: | Amino acids profile of Lannea microcarpa seed kernel (g/100 g protein) |
 | |
| *Essential amino acids, TEAA: Total essential amino acids, TNEAA: Total non-essential amino acids | |
| Table 4: | Amino acids score for Lannea microcarpa seed kernel |
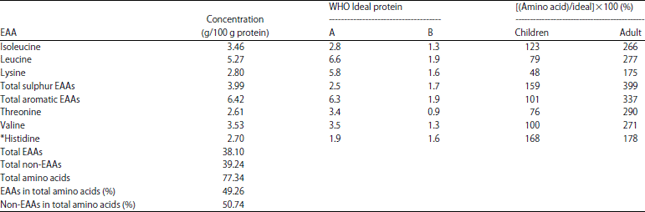 | |
| *Essential for children, A: WHO/FAO/UNU ideal protein for preschool children aged 3-10 years, B: WHO/FAO/UNU ideal protein for adult, EAAs: Essential amino acids | |
Viscosity is an important parameter used in assessing the quality of oil. The value obtained 32.35±3.11 cSt at 40°C and is lower compared to 33.1 cSt at 40°C for Adansonia digitata seed oil reported by Warra et al.19.
The refractive index is used to assessed oil contamination and adulteration20. The value recorded was 1.48±0.20 at 40°C and is relatively lower than 1.47 for Adansonia digitata seed oil19. The value obtained in this study is within the permissible range of 1.478-1.479 set as standard refractive index of vegetable oil21.
The saponification value of the oil was 192.54±3.78 mg KOH g–1 of oil. The value is lower than 194.23 mg KOH g–1 oil but higher compared to 156 mg KOH g–1 for Lannea microcarpa and Adansonia digitata seed oil, respectively18,19. Saponification value is used in assessing the degree of adulteration. The high saponification value of the oil indicates its suitability for cosmetic production22.
The peroxide value is an important parameter for oxidative rancidity in oils and fats, this is a measure of concentration of peroxides and hydro peroxides formed in the initial stage of lipid oxidation. The peroxide value obtained was low (1.57±0.11 meq of O2 kg–1 of oil). The recorded value is in agreement with 1.48 meq of O2 kg–1 oil for Lannea microcarpa seed oil18. Low peroxide value obtained is an indication that the oil is not likely to be liable to oxidative rancidity at room temperature23.
The acid value is used to indicate the level of rancidity of oils (edibility) and their suitability in paint and soap industry24. The acid value of the oil was 1.50±0.41 mg KOH g–1 of oil, which is higher than 1.21 mg KOH g–1 for Lannea microcarpa seed oi and 0.47 mg KOH g–1 for Adansonia digitata seed oil18,19. The low acid value suggested that the oil is good for human consumption since it falls within the range of 0.00-3.00 mg KOH g–1 recommended for cooking oil and consequently, is not suitable for soap production with respect to its acid value25,26.
Iodine value is the measure of the degree of unsaturation in oil or fat. Iodine value is a useful parameter in studying oxidative rancidity and chemical stability properties of different oil27, also used for determining the level of oxidative deterioration of the oil by enzymatic or chemical oxidation28. The iodine value of the oil was 60.13±4.84 g of I2/100 g of oil, which is relatively lower than that of Lannea microcarpa seed oil (61.33 g of I2/100 g) reported by Bazongo et al.18, but higher than 23.03 g of I2/100 g for Adansonia digitata seed oil19. The high iodine value indicates that the oil is edible and has high content of unsaturation (C=C double bond), suggesting the usefulness of the oil in domestic and industrial applications. Oil with iodine value less than 1.30 is reported to be non-drying oil and is therefore not suitable for making paint28.
Fatty acids composition: The result of fatty acids content of Lannea microcarpa seed oil is presented in Table 2. The result obtained shows the presence of both saturated and unsaturated fatty acids in the seed oil with saturated fatty acids accounting to 27.95% of the total fatty acids content, out of which stearic acid (11.35±1.20%) and palmitic acid (15.56±2.15%) are pre-dominant. On the other hand, the unsaturated fatty acids amounted to 72.05% of the total fatty acids content in which 45.33% is the total monounsaturated fatty acids and 26.72% is that of polyunsaturated fatty acids. Oleic acid (44.36±3.23%), linoleic acid (14.27±1.17%) and arachidonic acid (12.45±2.76%) are the pre-dominant unsaturated fatty acids present in the seed oil. The values obtained are in agreement with the result obtained for Lannea microcarpa seed oil18, but with slight variations which may be attributed to the seed maturity, climatic conditions, growth location and interactions between these factors. Ajayi29 reported similar results for Adansonia digitata seed oil. Oleic and palmitic acids have been reported to be effective percutaneous absorption enhancers while linoleic acid is the most frequently used fatty acid in cosmetic products as it moisturizes the skin, aids in the healing process of dermatoses and sunburns30. The result suggested the potential use of Lannea microcarpa seed oil in cosmetic industries in addition to its domestic use as cooking oil. However, the presence of linoleic acid in appreciable amount indicates that the oil is nutritious and may be use in alleviating fatty acid, deficiency syndrome and in preventing coronary heart disease29.
Amino acids composition: Table 3 showed the amino acids profile of Lannea microcarpa seed kernel. Twenty amino acids are commonly found as components of proteins31. Seventeen amino acids were detected as a result of the conversion of glutamine and asparagine to glutamic and aspartic acids, respectively and complete destruction of tryptophan during acid hydrolysis32,33. The result indicated that non-essential amino acids are higher in concentration and accounted for 50.74% compared to the essential amino acids which constitute 49.26% of the total amino acids analysed.
Among the essential amino acids, arginine and leucine are the predominant amino acids, while glutamic, aspartic and aspartic acids are the major non-essential amino acids in Lannea microcarpa seed kernel. The result obtained agree with that reported for Adansonia digitata and Melon seeds6.
Ideally, the protein in the diet should provide the body’s requirement of all the essential amino acids, in the same relative proportions. The contents of essential amino acids in Lannea microcarpa seed kernel are compared with the amino acid requirement stated by the WHO for adults and children aged 3-10 years and the result presented WHO/FAO/UNU34 in Table 4. The result shows that all the essential amino acids exceeded the reference value for adults. Similarly, isoleucine, valine, histidine, sulphur containing amino acids (methionine and cysteine), total aromatic essential amino acids (phenylalanine and tyrosine) exceeded the reference value for preschool children. However, for preschool children, lysine, leucine and threonine are below the standard requirement for children with lysine as the most limiting essential amino acid. This implies that, if properly utilized the kernel of Lannea microcarpa seed could be a good supplement of protein.
The results of the present study revealed that the seed kernel of Lannea microcarpa contain an appreciable amounts of essential fatty acids of which oleic and linoleic acids are the predominant unsaturated fatty acids. The oil therefore have the potential for use as vegetable oil, food, pharmaceutical and industrial application. The seed kernel also contains an appreciable amount of amino acids in which glutamic and aspartic acids dominated the non-essential component, while leucine and isoleucine dominate the essential component of the amino acids profile. The seed of Lannea microcarpa if properly utilized could be a potential supplement of raw materials for food, cosmetics and pharmaceutical industries.