Research Article
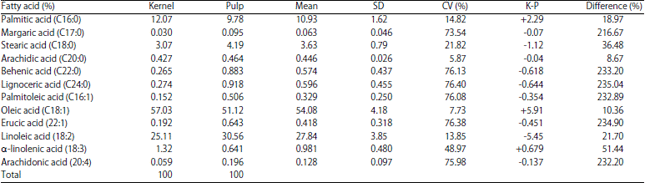
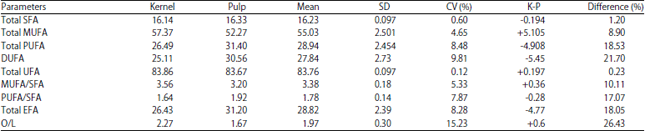
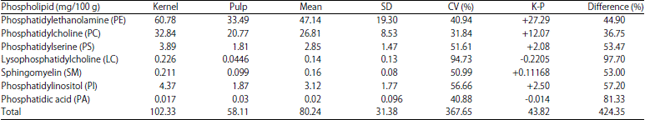
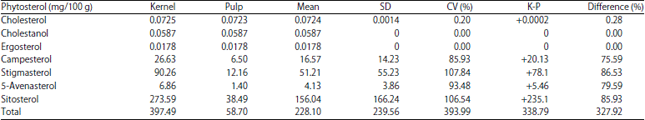
Comparative Studies on the Lipid Profile of Shea (Vitellaria paradoxa C.F. Gaertn.) Fruit Kernel and Pulp
Department of Chemical Sciences, Federal University Wukari, PMB 1020, Taraba State, Nigeria
LiveDNA: 234.1611
Chrysanthus Andrew
Electroplating and Metal Finishing Division, Central Electrochemical Research Institute, 630003, Karaikudi Tamil Nadu, India
Rasaq Bolakale Salau
Department of Chemistry, Federal University of Technology, Minna, Niger State, Nigeria
Benevolent Orighomisan Atolaiye
Department of Chemistry, Nasarawa State University, PMB 1022, Keffi, Nigeria
Gary Garbunga Yebpella
Department of Chemical Sciences, Federal University Wukari, PMB 1020, Taraba State, Nigeria
Michael Okey Enemali
Department of Biochemistry and Molecular Biology, Nasarawa State University, PMB 1022, Keffi, Nigeria