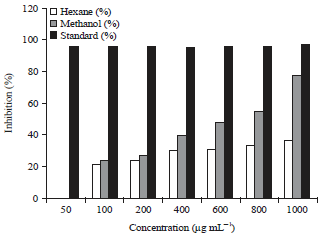
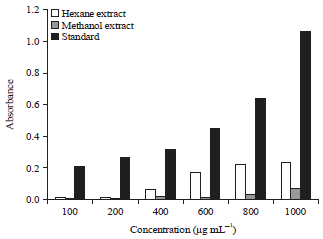
Research Article
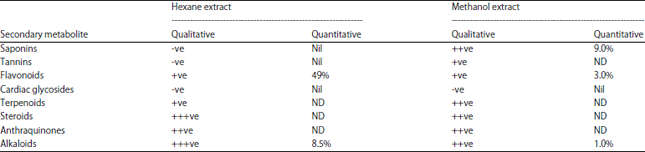
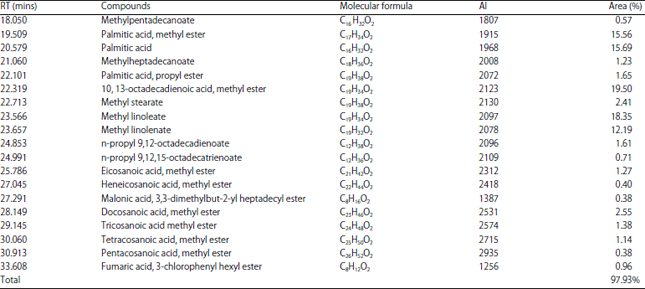
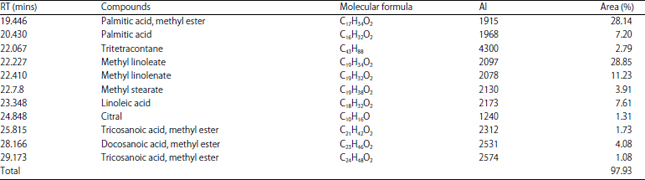
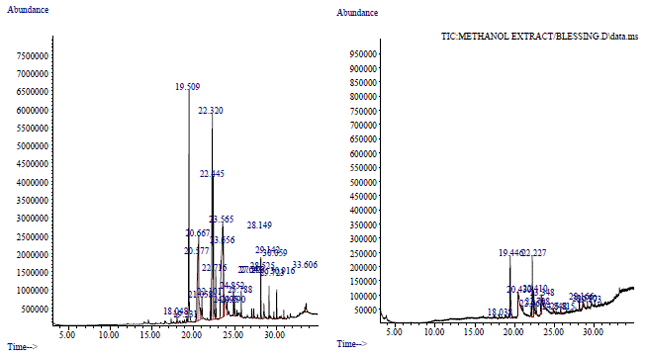
Effect of solvent on Chemical Composition and Antioxidant Activities of Abelmoschus esculentus (L.) Moench Pods’ Oils Extracts
Natural Products/Medicinal Chemistry Unit, Department of Chemistry, University of Ibadan, Nigeria
LiveDNA: 234.19450
Blessing H. Ojeyinka
Department of Chemistry, National Open University of Nigeria, Ibadan Study Center, Ibadan, Nigeria