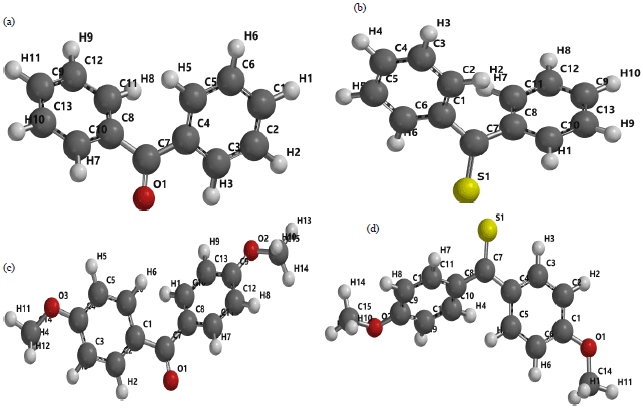
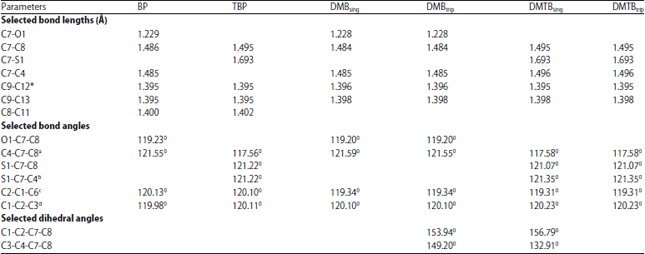
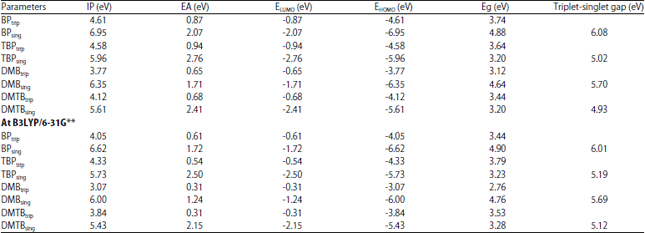
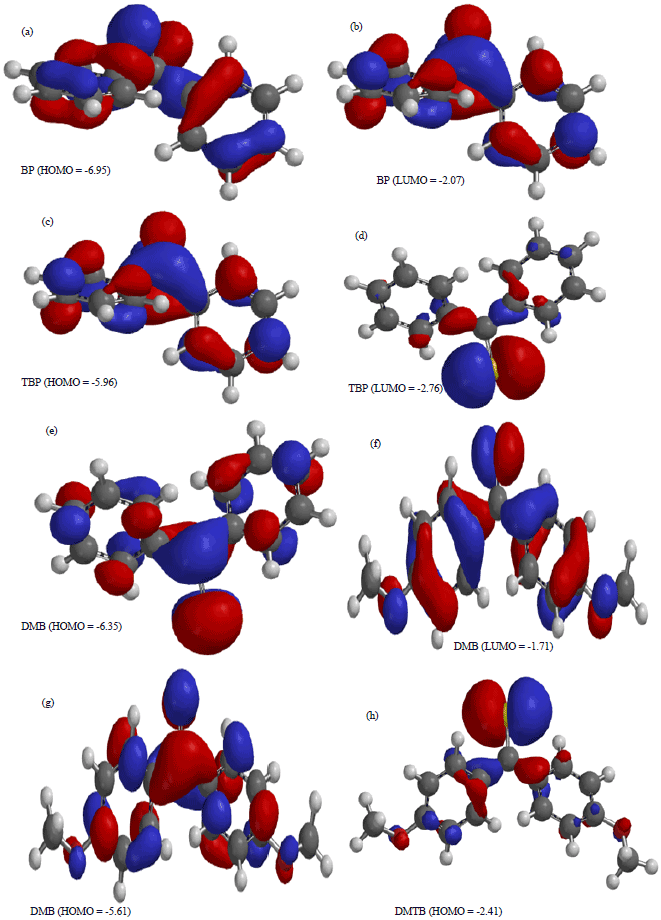
Research Article
Nature of Ground State of Benzophenone and some of its Substituted Derivatives: Experimental and DFT Study
Department of Pure and Industrial Chemistry, University of Nigeria, Nsukka, Nigeria
J. Adegboyega
Department of Chemistry, University of Ibadan, Nigeria
O.D. Okagu
Department of Pure and Industrial Chemistry, University of Nigeria, Nsukka, Nigeria
B.B. Adeleke
Department of Chemistry, University of Ibadan, Nigeria