ABSTRACT
Background: Salinization of land is one of the most widely serious problems in agriculture production. Using of exogenous bioactive substances not only improve the biological properties of several agricultural crops but also help in promoting their yield in saline environments. Methodology: In this study, the potential effect of crude extract of five marine algal species on the germination, growth and some enzymatic activities of salinity stressed wheat seedlings were investigated. Results: Increasing of salinity showed a sharp inhibition on the germination percent and seedling growth criteria of wheat seedlings. Algal presoaking of salinity stressed grains demonstrated a highly significant enhancement in the percentage of seed germination and growth parameters especially with the extract of red alga Laurencia obtusa. A marked promotion in the activity of the antioxidant enzymes, catalase (CAT) and superoxide dismutase (SOD) was recorded in the salinity stressed plants. The occurrence of bioactive compounds in algal extracts such as flavonoids, proline, ascorbic acid, citric acid and plant hormones could potentially participate in the alleviation of salinity stress. Conclusion: The present study confirmed the presoaking technique of marine algal extract is an effective technology in solving one of the greatest economic problems in agriculture.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2016.388.394
URL: https://scialert.net/abstract/?doi=jas.2016.388.394
INTRODUCTION
Soil salinity is one of the major environmental stresses that drastically reduce crop productivity around the world by reducing the crop growth and yield1,2. The loss of fertile agriculture land is predicted to increase globally within the next 25 years and reaches to 50% by the year 2050 due to the demand of intensive irrigation. Consequently, millions of hectares of usable land will become unsuitable for plant cultivation due to the continuous heighten of soil salinity3.
The adverse effects of salinity on crop growth include alteration of physiological and biochemical processes then causes modification in morphological and anatomical features4. Seed germination is the most sensitive part in the life cycle of crop plants and is a major limiting factor for upgrading wheat plants, like other crops, under saline conditions5,6.
Different approaches have been considered to maximize crop growth and productivity under salinity stress. The essential trend is to develop salt tolerance plants through genetic means. However, the genetic production of salinity tolerance cultivars is a long-term effort with inherent difficulties and complications. An alternative and simpler approach is to originate salinity tolerance plant through application of exogenous growth-promoting compounds2.
Marine algae possess abundant ecological diversity and synthesize metabolites with various structures and interesting activities to several biotechnological purposes7,8. Marine algae are classified based on their pigmentation into three groups: Chlorophyta (green algae), Phaeophyta (brown algae) and Rhodophyta (red algae). Some marine algae live in extreme environmental conditions that lead to the production of free radicals and other oxidizing agents. Therefore, their cells possess strategies for rapid formation of secondary metabolites, which protect them against harsh conditions9.
Thus, it is urgent to select the potential of marine algal extract for enhancing growth of salinity stressed crop plant and applying this technology for reusing of huge saline lands for sustainable agriculture purposes. However, the effect of marine algal extracts on germination and growth of wheat seedlings grown under salt stress conditions has yet not been studied. The present study was intended to investigate the efficiency of various marine algal extracts to stimulate the growth and metabolic activities of wheat seedlings grown under different salinity stress.
MATERIALS AND METHODS
Collection and preparation of marine algal species: The marine algae Caulerpa racemosa and Padina pavonica (Fig. 1a, b) were collected from the Mediterranean Sea at Abo Keer coast in Egypt, during spring season (April, 2015).
 | |
| Fig. 1(a-e): | Collected marine algal species, (a) Caulerpa racemosa, (b) Padina pavonica, (c) Sargassum muticum, (d) Galaxaura obtusata and (e) Laurencia obtusa |
While, the marine algae Sargassum muticum, Galaxaura obtusata and Laurencia obtusa (Fig. 1c-e) were collected during winter season (January, 2015) from the Red Sea at Hurghada coast in Egypt. All algae were washed with tap water and distilled water to remove impurities. The fresh seaweed samples were homogenised in distilled water (1:1 w/v) then filtered through Whatman No. 1 filter paper. This liquid extract was taken as 100% concentration. From the latter extract, 10% algal extract was prepared using distilled water.
Investigated crop plant: Pure identified strain of wheat plant was chosen for this study namely Triticum vulgare (var.6h Shandawel). Grains of the selected plant were obtained from the Ministry of Agriculture, Giza, Egypt.
Experimental design: A homogenous lot of grains of tested plant were surface sterilized by soaking in 0.01% HgCl2 solution for 3 min. Then the sterilized 20 seeds were presoaking in distilled water "Control" and different algal extracts for 18 h. Thereafter the grains were allowed to drain for one hour on stainless steel screen and then placed on filter paper for 24 h at normal room temperature and humidity to dry. The grains were transferred to sterile petri dish (15 cm diameter) containing water moisted filter paper. The seeds were placed in darkness to germinate at 25°C. Petri dishes were watered with various concentrations of NaCl (0, 10, 50, 100 and 150 mmol) for 6 days. At the end of the experimental period, the plumule and radical length, seedling fresh weight, carbohydrate and protein content in addition to antioxidant enzymes activities were revealed.
Biochemical analysis: The amino acids were determined according to Pellet and Young10. For each algal sample, 1.0 g was defatted using diethyl ether and hydrolyzed using 6N HCl. Aftre 24 h, the hydrochloric acid was evaporated on water bath at 50-60°C. The sample was dried till the formation of dry film. The sample was dissolved in sodium acetate buffer (pH 2.2) then the sample was injected to the amino acid analyzer (LC 3000 Eppendorf, Desert Research Center) for determination of amino acid composition.
Minerals concentrations were determined according to Allen et al.11. For vitamins, 5.0 g fresh sample was homogenized in 10 mL methanol and filtrate. The filtrate was extracted with diethyl ether using separating funnel. The upper layer was injected to the HPLC analyzer (DAD 3000 Germany, Desert Research Center). For phenolic compound, filtrated methanolic sample was injected to the HPLC analyzer. For plant hormones, methanolic sample was stored in freezer for 24 h. After evaporation of methanol, algal sample was extracted by ethyl acetate then introduced to the HPLC analyzer operating at 254 nm. Radial-Pack A Cartilage C18 (100×8 mm) column and Z-module redial compression system were used.
In case of enzyme activity, samples of plant tissues (0.5 g) were homogenized with 0.1 M phosphate buffer (cold) and then centrifuged for 15 min. Superoxide dismutase (SOD) activity was estimated according to Beyer and Fridovich12 and catalase (CAT) activity was estimated according to Aebi13.
Statistical analysis: All experiments were achieved using a randomized design. The data are presented as the Mean±SE of three replicates. The significance between algal treated and control seedlings were analyzed using the Student’s t-test taking p≤0.05 as significant.
RESULTS
Change in percentage of germination: The percentages of germination of wheat grains presoaked in 5 algal extracts and treated with different concentrations of NaCl were demonstrated in Fig. 2. Presoaking of wheat grains in algal extracts recorded a significant increase in percentage of germination by about 12 and 25% when compared with control (water presoaked grains). Obviously, an inversely relationship between salinity and germination percentage was recorded.
 | |
| Fig. 2: | Effect of salinity on germination percentage of wheat grains presoaked in different marine algal extracts. Data are the mean of three replicates and error bars represent the standard errors of the means |
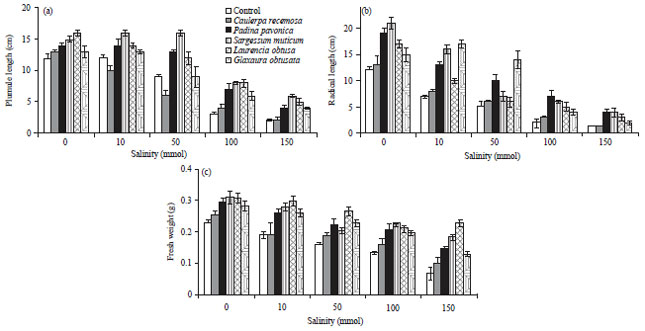 | |
| Fig. 3(a-c): | Effect of salinity on growth of wheat seedlings presoaked in different marine algal extracts, (a) Plumule length, (b) Radical length and (c) Fresh weight. Data are the mean of three replicates and error bars represent the standard errors of the means |
It is apparent that germination percentage was significantly decreased with increasing the concentrations of NaCl by about 37 and 49% at the higher salinity levels (100 and 150 mmol NaCl). However, presoaking of wheat grains in algal extracts of C. racemosa, P. pavonica, S. muticum, L. obtusa and G. obtusata progressively alleviated the repression effect of NaCl stress by increasing the germination percentage with values of 25, 38, 66, 96 and 71%, respectively.
Change in growth parameters: Figure 3a showed that the wheat seedlings treated with different concentrations of NaCl recorded a significant decrease in their growth parameters (plumule length, radical length and seedling fresh weight) when compared with unstressed seedlings.
The seedlings developed from the algal extract presoaking recorded a highly significant increase in all tested growth parameters when compared with water presoaking seedlings. Moreover, presoaking of wheat grains in various algal extracts reduced the repression effect of salinity stress by improving all growth parameters compared with water presoaking seedlings "Control". A dramatic increase in these parameters was manifested 16-200% for plumule length, 14-300% for radical length and 12-240% for seedling fresh weight. In general, the maximum increase in seedling plumule and radical length was obtained in case of seedlings treated with S. muticum extract. Whereas, L. obtusa extract stimulated a highly significant increase in seedling fresh weight.
Change in carbohydrate and protein metabolism: Figure 4a shows that the carbohydrate content in wheat seedlings. Generally, the carbohydrate content of seedlings decreased with increasing salinity levels, in a concentration dependent manner, in case of unalgal treated seedlings. However, presoaking of wheat grains in different algal extracts stimulate the accumulation of carbohydrate in seedlings treated with different salinity levels with mean values ranged from 15-139% when compared with control seedlings. The maximum increment in carbohydrate content was obtained with seedlings treated with L. obtusa extract at 100 mmol salinity level.
The total protein content of wheat seedlings grew in different salinity levels (Fig. 4b) was lower than the corresponding control and the protein content of seedlings decreased with increasing salinity levels up to 150 mmol. Presoaking of wheat grains in different algal extracts relieved the effect of NaCl stress by increase the seedlings total protein content by 10-56% when compared with control. The maximum increment was obtained with red alga L. obtusa extract at different salinity levels.
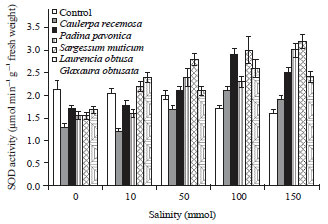
Change in antioxidant enzymatic activities: Figure 5a showed that the catalase (CAT) activity of wheat seedlings was decreased with increasing salinity level up to 150 mmol NaCl. Moreover, presoaking of wheat grains in different algal extracts showed a sharp increase in seedling CAT activity with increasing salinity. The maximum increase in CAT activity was obtained with extract of red alga L. obtusa at salinity level 50 mmol NaCl.
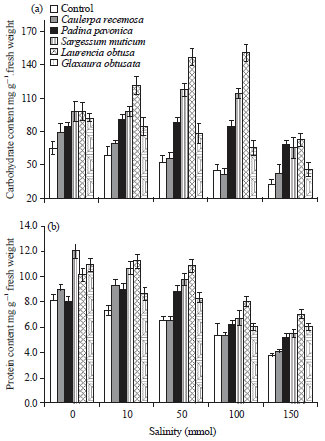 | |
| Fig. 4(a-b): | Effect of salinity on metabolic activity of wheat seedlings presoaked in different marine algal extracts, (a) Carbohydrate content and (b) Protein content. Data are the mean of three replicates and error bars represent the standard errors of the means |
Concerning the superoxide dismutase (SOD) activity. Figure 5b shows that SOD activity of wheat seedlings grown under different salinity levels was lower than unstressed seedlings. It is worth to mention that the SOD activity of seedlings treated with different algal extracts showed a sharp increase in SOD activity with increasing salinity especially under higher NaCl concentrations (100-150 mmol). Also, the maximum increase in SOD activity was achieved with extract of red alga L. obtusa at higher salinity levels.
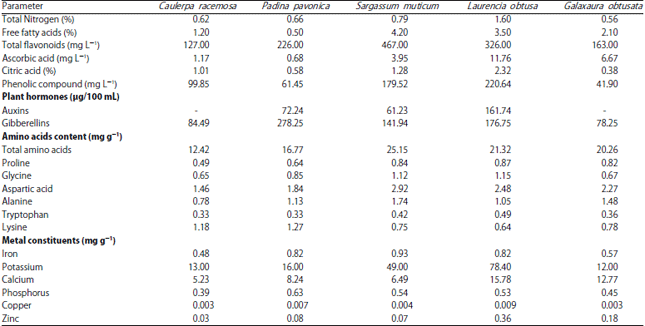
Characterization of marine algal extracts: Table 1 shows that the total nitrogen content of red alga L. obtusa extract was higher than that found in all algae, while the lowest content was recorded in extract of red alga G. obtusata. Similar trend was detected in content of phenolic compound, citric acid, potassium and copper with different algal extracts. At the same time, the highest content of ascorbic acid, proline and tryptophan was recorded in algal extract of red alga L. obtusa whereas the lowest level was recorded in extract of green alga C. racemosa. Also, Table 1 demonstrated that the algal extract of brown alga S. muticum contained the highest content of iron, total amino acids and total flavonoids, while extract of green alga C. racemosa contained the lowest content. Analysis of phytohormones revealed that, the highest auxins content was recorded in L. obtuse extract (161.74 μg/100 mL) followed by P. pavonica extract (72.24 μg/100 mL) and the absence of auxins in C. racemosa and G. obtusata extracts. Also, the gibberellins content of P. pavonica extract (278.25 μg/100 mL) was higher than that found in other marine algal extracts, while the lowest content was recorded in extract of red alga G. obtusata (78.25 μg/100 mL).
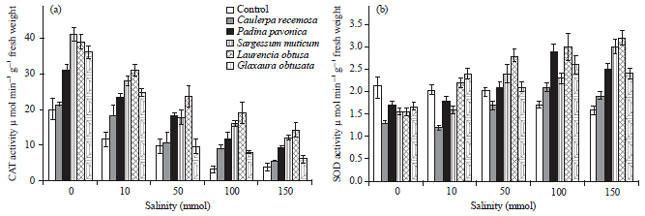 | |
| Fig. 5(a-b): | Effect of salinity on antioxidant enzymatic activities of wheat seedlings presoaked in different marine algal extracts, (a) Catalase and (b) Superoxide dismutase. Data are the mean of three replicates and error bars represent the standard errors of the means |
| Table 1: | Some biochemical analysis of different marine algal species |
 | |
DISCUSSION
Soil salinity is one of the major abiotic stresses that are potentially deleterious to the crop growth and productivity. The inhibitory effects of salinity could be attributed to the oxidative damage of salinity which led to the generation of Reactive Oxygen Species (ROS), inhibition of antioxidant systems or energy expenditure during osmotic adjustment to salinity14-16.
The obtained data showed a significant decrease in the germination percentage of wheat grains at different salinity levels compared with water presoaked grains. The adverse effect of NaCl has been attributed to change in permeability of plasma membranes and facilitate the accumulation of toxic ions to embryo6,17. Presoaking of grains in different marine algal extracts significantly increased the germination of wheat plant under NaCl stress. However, the most effective algal extract, which had the highest germination percentage was recorded with L. obtusa extract. This increment could be due to the presence of some mineral constituents such as Ca+2 in L. obtusa extract than other algal extracts, which reduce the adverse effect salinity on germination process by inhibition of Na+ uptake6.
Salinity may affect seedling growth by lowering the absorption of water and/or decreasing the level of gibberellins in germinating seeds17. Consistent with this, the application of different marine algal extracts had a significant stimulatory effect on the growth parameters of wheat seedlings under salinity stress. This result could be attributed to the existence of some bioactive compounds as ascorbic acid, auxins and gibberellins in marine algal extract which mitigate the adverse effect of salinity on growth of wheat seedlings18-20. Also, the presence of inorganic phosphate in marine algal extract plays a vital role in energy transfer, growth and protecting wheat seedlings from salt toxicity1.
Antioxidant enzymes are the first response mechanism against salinity stress. Therefore, these enzymes are important for the achievement of seedling growth. Seedling cells have several protective mechanisms against ROS production that take place during germination stage, but they seem to be disturbed by salinity17. The present results revealed significant decrease in the activities of CAT and SOD in salinity stressed seedlings. At the same time, presoaking of grains in marine algal extract, especially L. obtusa, increasing the activity of seedling antioxidant enzymes which are involved in the scavenging of active oxygen species6,21. The increment in the enzyme activities could be attributed to the presence of the antioxidant and osmoprotectant components, such as proline, phenolic compounds, fatty acids and vitamins in the investigated marine algal extracts.
CONCLUSION AND FUTURE RECOMMENDATION
Marine macroalgae originate a variety of highly bioactive metabolites, with structures that cannot be found in other organisms. Although, many products obtained from algae have economic importance and are broadly used, the present study is the first report, which demonstrates the application of five marine algal extracts to overcome the deleterious effect of salinity on wheat seedlings.
Our results confirmed that presoaking of wheat grains in algal extracts especially that of red alga L. obtusa, increases the germination percentage, growth and antioxidant content of salinity stressed seedlings. Consequently, study should continue to isolate the main active compounds that could be used in alleviation of salinity and determine the efficiency of these materials under natural field condition.
ACKNOWLEDGMENTS
The facilities provided by the Faculty of Science, Damanhour University, Egypt are greatly acknowledged and appreciated.
REFERENCES
- Singh, J.S., V.C. Pandey and D.P. Singh, 2011. Efficient soil microorganisms: A new dimension for sustainable agriculture and environmental development. Agric. Ecosyst. Environ., 140: 339-353.
CrossRefDirect Link - Karlidag, H., E. Yildirim and M. Turan, 2011. Role of 24-epibrassinolide in mitigating the adverse effects of salt stress on stomatal conductance, membrane permeability and leaf water content, ionic composition in salt stressed strawberry (Fragaria χ ananassa). Scientia Hortic., 130: 133-140.
CrossRefDirect Link - Spano, C. and S. Bottega, 2016. Durum wheat seedlings in saline conditions: Salt spray versus root-zone salinity. Estuarine Coastal Shelf Sci., 169: 173-181.
CrossRefDirect Link - Tamayo-Ordonez, M.C., L.C. Rodriguez-Zapata, J.A. Narvaez-Zapata, Y.J. Tamayo-Ordonez, B.A. Ayil-Gutierrez, F. Barredo-Pool and L.F. Sanchez-Teyer, 2016. Morphological features of different polyploids for adaptation and molecular characterization of CC-NBS-LRR and LEA gene families in Agave L. J. Plant Physiol., 195: 80-94.
CrossRefDirect Link - Wang, C.L., S.C. Zhang, S.D. Qi, C.C. Zheng and C.A. Wu, 2016. Delayed germination of Arabidopsis seeds under chilling stress by overexpressing an abiotic stress inducible GhTPS11. Gene, 575: 206-212.
CrossRefDirect Link - Zehra, A., B. Gul, R. Ansari and M.A. Khan, 2012. Role of calcium in alleviating effect of salinity on germination of Phragmites karka seeds. South Afr. J. Bot., 78: 122-128.
CrossRefDirect Link - Torres, F.A.E., T.G. Passalacqua, A.M.A. Velasquez, R.A. de Souz, P. Colepicolo and M.A.S. Graminha, 2014. New drugs with antiprotozoal activity from marine algae: A review. Rev. Bras. Farmacogn., 24: 265-276.
CrossRefDirect Link - Dore, C.M.P.G., M.G.C.F. Alves, L.S.E.P. Will, T.G. Costa and D.A. Sabry et al., 2013. A sulfated polysaccharide, fucans, isolated from brown algae Sargassum vulgare with anticoagulant, antithrombotic, antioxidant and anti-inflammatory effects. Carbohydr. Polym., 91: 467-475.
CrossRefDirect Link - Gonzalez, A.E.B., M.B. Charles, J. Mancini-Filho and A.V. Novoa, 2009. Seaweeds as sources of antioxidant phytomedicines. Rev. Cubana Plant Med., 14: 1-18.
Direct Link - Beyer, Jr. W.F. and I. Fridovich, 1987. Assaying for superoxide dismutase activity: Some large consequences of minor changes in conditions. Anal. Biochem., 161: 559-566.
CrossRefDirect Link - Aebi, H., 1984. Catalase in vitro. In: Methods in Enzymology, Packer, L., Academic Press, Cambridge, Massachusetts, United States, ISBN: 9780121820053, pp: 121-126.
CrossRefDirect Link - Filippou, P., P. Bouchagier, E. Skotti and V. Fotopoulos, 2014. Proline and reactive oxygen/nitrogen species metabolism is involved in the tolerant response of the invasive plant species Ailanthus altissima to drought and salinity. Environ. Exp. Bot., 97: 1-10.
CrossRefDirect Link - He, Y., J. Yang, B. Zhu and Z.J. Zhu, 2014. Low root zone temperature exacerbates the ion imbalance and photosynthesis inhibition and induces antioxidant responses in tomato plants under salinity. J. Integr. Agric., 13: 89-99.
CrossRefDirect Link - Chakraborty, K., D. Bhaduri, H.N. Meena and K. Kalariya, 2016. External potassium (K+) application improves salinity tolerance by promoting Na+-exclusion, K+-accumulation and osmotic adjustment in contrasting peanut cultivars. Plant Physiol. Biochem., 103: 143-153.
CrossRefDirect Link - Fercha, A., A.L. Capriotti, G. Caruso, C. Cavaliere, R. Samperi, S. Stampachiacchiere and A. Lagana, 2014. Comparative analysis of metabolic proteome variation in ascorbate-primed and unprimed wheat seeds during germination under salt stress. J. Proteomics, 108: 238-257.
CrossRefDirect Link - Ye, N. and J. Zhang, 2012. Antagonism between abscisic acid and gibberellins is partially mediated by ascorbic acid during seed germination in rice. Plant Signal. Behav., 7: 563-565.
CrossRefDirect Link - Leitaoa, A.L. and F.J. Enguita, 2016. Gibberellins in Penicillium strains: Challenges for endophyte-plant host interactions under salinity stress. Microbiol. Res., 183: 8-18.
CrossRefDirect Link - Nassar, R.M.A., N.T. Shanan and F.M. Reda, 2016. Active yeast extract counteracts the harmful effects of salinity stress on the growth of leucaena plant. Scientia Hortic., 201: 61-67.
CrossRefDirect Link - Rentel, M.C. and M.R. Knight, 2004. Oxidative stress-induced calcium signaling in Arabidopsis. Plant Physiol., 135: 1471-1479.
Direct Link








