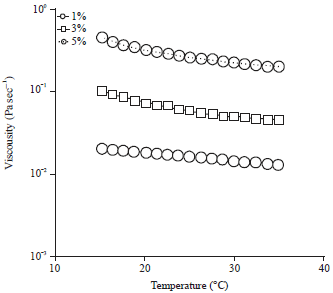
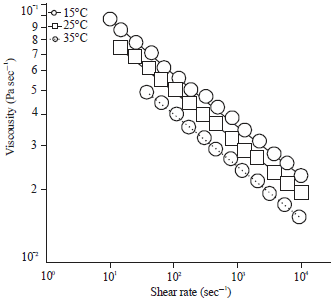
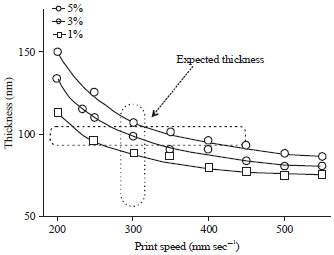
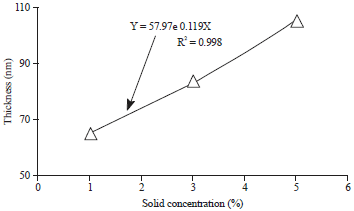
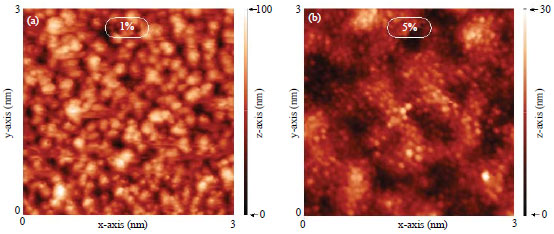
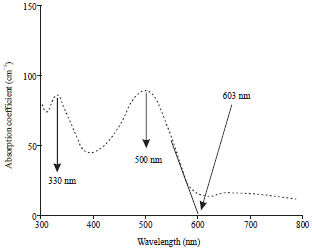
Research Article
MEH-PPV: PCBM Solution for Screen-printing Technique in Polymer Solar Cell Fabrication
Faculty of Engineering, Physics and Nanotechnology, VNU-University of Technology and Engineering, No. 144, Xuan Thuy Str., Cau Giay Distr., Hanoi, Vietnam
Dinh Van Thuong
Faculty of Engineering, Physics and Nanotechnology, VNU-University of Technology and Engineering, No. 144, Xuan Thuy Str., Cau Giay Distr., Hanoi, Vietnam