Research Article
Chemical Composition and Antimicrobial Activities of Cyanobacterial Mats from Hyper Saline Lakes, Northern Western Desert, Egypt
National Institute of Oceanography and Fisheries, El-Kasr El-Eny St., 101, Cairo, Egypt
Cyanobacteria were found to be a rich source for various products of commercial, pharmaceutical or toxicological interest. They can produce primary metabolites, such as proteins, fatty acids, vitamins or pigments (Borowitzka, 1988, 1995) and many secondary metabolites with different antifungal and antibacterial activities (El Semary, 2012a, b; Itoh et al., 2014). Although cyanobacteria are considered as a prolific source of secondary metabolites with a wide spectrum of bioactive effects (Thajuddin and Subramanian, 2005), they are still not thoroughly investigated and have been poorly exploited (Ehrenreich et al., 2005; Barrios-Llerena et al., 2007).
Many investigations have focused on different cyanobacterial species in pure culture as a prolific source for different antimicrobial metabolites; Hormothamnion enteromorphoides (Gerwick et al., 1989), Mycobacterium tuberculosis (Rao et al., 2007), Nostoc insulare (Volk, 2007), Anabaena sp. (Chauhan et al., 2010), Leptolyngbya sp. and Phormidium sp. (El Semary, 2012a, b) and Nostoc commune (Itoh et al., 2014). More recently, cyanobacteria in dens mats was found to have the ability to produce secondary metabolites with antibacterial, antidiatom and quorum-sensing inhibitory compounds under in situ conditions (Dobretsov et al., 2011). Cyanobacterial mats have the ability to produce dens mats in hot springs, Antarctic lakes, deep seas and hyper saline marches (Fenical and Jensen, 2006; Biondi et al., 2008).
In extreme conditions, extremophile mats are composed of different physiological groups of microbes such as cyanobacteria, photoheterotrophic, chemoautotrophic and heterotrophic organisms (Van Gemerden, 1993; Stal, 1995). The bioactivity of mixed microbial communities has advantage of microbial relationships in natural communities that do not exist in pure cultures. They are believed to be a rich source of bioactive metabolites (Burja et al., 2001; Gerth et al., 2003; Dahms et al., 2006). Furthermore, there is a possibility to isolate novel compounds produced by microorganisms that are difficult to cultivate in the laboratory. Such an approach provides not only increased possibilities to discover more versatile secondary metabolites but also increases our understanding of the chemical environment and cell to cell relationship in mixed microbial communities (Abed et al., 2013). Additionally, microbial composition of antibiotic producing and nonproducing cyanobacterial mats were significantly different, suggesting that different microorganisms of the studied mats might have produced different types of chemical compounds (Abed et al., 2011).
This study aims at studying the antimicrobial activities of dens growing cyanobacterial mats collected from hyper saline lakes of the northern western desert, Egypt and screening the chemical compounds produced by these microbial mats.
Samples collection: Cyanobacterial mats were collected from Zieton, Aghormy and Maraqi lakes in Siwa city, northern western desert, Egypt (Fig. 1) during mid-spring in 2014. The lakes main features and characters were presented in details by Abd El-Karim and Goher (2016). From each lake, approximately 1 kg of the cyanobacterial mats (1-3 mm top layer) were collected from the margins of each lake, transferred on ice to the laboratory and stored at-30°C. Few days later, the mats were used for antimicrobial activities and chemical composition analysis.
Preparation of extracts: Cyanobacterial mats were dried under forced-air circulation at 38±2°C until its weight stabilized. Fifteen gram dry weight of the cyanobacterial mat was sonicated (apparatus name) for 15 min in 60 mL of ethyl acetate, petroleum ether, methanol, ethanol, acetone, chloroform and n-hexane, then left overnight. All obtained extracts were filtered on a Whatmann No. 1 filter paper. Extracts were evaporated under vacuum till dryness. After complete dryness, the extracts were weighted and dissolved in dimethyl sulphoxide (DMSO) to obtain concentrations of 1 mg mL–1. Later, extracts of different solvents with concentrations of 5 mg mL–1 were prepared to test the resistant organisms for lower concentrations of 1 mg mL–1. Extracts were stored in glass vials in dark at -30°C. All the crude extracts were used for the antimicrobial screening assays with varying concentrations.
Tested microorganisms: Gram negative bacterial strains; Escherichia coli (E. coli), Pseudomonas aeruginosa (P. aeruginosa), Vibreo cholerae (V. cholerae), Shigella sp., Aeromonas sp. and Flavobacterium sp. and Gram positive bacterial strains; Staphylococcus aureus (S. aureus) and S. lentus and yeast like fungi; Candida albicans (C. albicans) and C. tropicalis were used for the study. The parent cultures were obtained from microbiology and parasitology departments and the subcultures were maintained once in 15 days.
Antimicrobial activity: Antimicrobial activity was screened using Mueller Hinton Agar, MHA, (Oxoid Ltd., Basing Stoke, Hampshire, England). The MHA plates were prepared by pouring 15 mL of molten media into sterile petri plates.
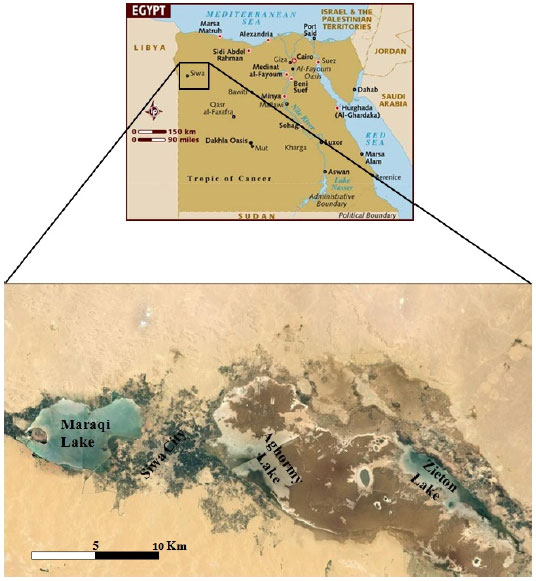 | |
| Fig. 1: | Map showing sampling lakes |
The plates were allowed to solidify for 5 min. A standardized bacterial inoculum was uniformly spread on the MHA agar surface using sterile cotton swabs. Immediately, wells of 5 mm in diameter were made on the agar surface using a sterile metallic cylinder. Next, 50 μL of each extract dissolved in DMSO was added into the wells and the plates were incubated at 37°C for 24 h. At the end of the incubation, the diameter of the inhibition zone formed around the wells was measured. A well containing only DMSO were used as negative control and all tests were done in triplicate.
Chemical composition of extracts: The extracts in DMSO were analyzed by a coupled gas chromatography-mass spectrometry (GC-MS). The separation of compounds and their analysis was performed using Agilent 7000 series Quadrupole GC-MS system with electron impact ionization. The total GC run time was 52 min and the carrier gas was helium. The initial oven temperature was held at 90°C for 1 min and then reached 300°C in 13 min, after which it was held at this temperature for 20 min. The injector temperature was 300°C. NIST MS spectral library and Agilent’s Retention Time Locked (RTL) databases was used to identify the compounds in the extracts. The closest match with the highest probability in the library was recorded.
Phytochemical screening: Cyanobacterial extracts were prepared as illustrated above in 50 mL of ethyl acetate, petroleum ether, methanol, ethanol, acetone, chloroform and n-hexane and the extracts were concentrated till reached to about 10 mL. The extracts were analyzed for the presence of phenolic compounds, flavonoids, terpenoids, saponins, alkaloids and glycosides (Raaman, 2006).
Phenolic compounds, ferric chloride test: Each extract (1 mL) was diluted with 5 mL of distilled water and few drops of 5% ferric chloride were added. Bluish black color indicated the presence of Phenolic compounds.
Flavonoid, alkaline reagent test: Few drops of sodium hydroxide were added into the extracts to give intense yellow color. The disappearance of color after addition of dilute hydrochloride acid showed the presence of flavonoid.
Terpenoids, Salkowski’s test: To 1 mL extract few millilitre of chloroform was added followed by concentrated sulphuric acid to form a layer. Reddish brown color at the interface indicated the presence of terpenoids.
Saponins, Froth test: Each extract (1 mL) was diluted with distilled water and made up to 5 mL. The suspension was vigorously shaken. Add some drops of olive oil. The formation of stable foam was taken as an indication for the presence of saponins.
Alkaloids, Wagner’s test: One milliliter of extracts was stirred with few ml of dilute hydrochloric acid and filtered. Then, few drops of Wagner’s reagent were added at the side of the test tube. The formation of reddish-brown precipitate showed the presence of alkaloids.
Glycosides, Keller-Kiliani’s test: Tested extract was treated with 2 mL of glacial acetic acid containing one drop of 5% ferric chloride followed by addition of 1 mL of concentrated sulphuric acid. A brown ring at interface is characteristic of cardenolide deoxy sugar. Lower reddish brown layer and upper acetic acid layer which turns bluish green would indicate a positive test for glycosides.
Statistical analysis: Differences between solvent control and the treatments were determined by one-way analysis of variance (ANOVA). Comparison of chemical extracts was carried out using the PRIMER 5 software package (Primer-E Ltd., Ivybridge, UK). For cluster analysis of chemical extracts Bray-Curtis similarities were used to produce a matrix based on the total number of peaks observed in all extracts and the presence or absence of these peaks in individual extracts.
Antimicrobial activity: The antimicrobial activities of cyanobacterial mats from three hyper saline lakes in seven organic solvents were assayed against eight bacterial strains and two yeast strains by evaluating the inhibition zones (Table 1). Generally, the 10 microbes showed differential response against the cyanobacterial extracts at the concentration of 1 mg mL–1. At this concentration, the growth of P. aeruginosa out of eight pathogenic bacteria (i.e., S. lentus, S. aureus, Flavobacterium sp., Shigella sp., P. aeruginosa, E. coli, V. cholera and Aeromonas sp.) and C. albicans out of two yeasts (C. albicans and C. tropicalis) showed no inhibition at different extracts. Staphylococcus aureus and Aeromonas sp. were the most susceptible bacteria to different extracts from different lakes.
| Table 1: | Antimicrobial activities around the wells (inhibition zone in diameter, mm) of different organic extracts (50 µ mL/well) of cyanobacterial mats from Aghormy, Zieton and Maraqi lakes at concentration of 1, 5 mg mL–1 |
 | |
| EA: Ethyl acetate, PE: Petroleum ether, ME: Methanol, E: Ethanol, A: Acetone, NH: n-hexane and CH: Chloroform | |
Extracts of cyanobacterial mats of Aghormy Lake were more effective against V. cholera which was inhibited by ethanol and ethyl acetate (polar extracts) and petroleum ether (non-polar extracts) also against C. tropicalis which was inhibited by ethyl acetate, petroleum ether, n-hexane and chloroform. Staphylococcus lentus, was inhibited by n-hexane and chloroform extracts of cyanobacterial mats from Zieton Lake. Escherichia coli, Flavobacterium sp. and Shigella sp. were inhibited by extracts from Zieton and Maraqi lakes, whereas they were not inhibited by any extracts from Aghormy Lake.
Cyanobacterial mats extracts with concentration of 5 mg mL–1 were found to be significantly effective (p<0.005) against the most resistant microbes, S. lintus, C. tropicalis, C. albicans and V. cholera, except for P. aeruginosa which was inhibited only by chloroform from Maraqi Lake and acetone from Zieton Lake. The widest mean inhibition zone of microbial strains was recorded by ethyl acetate (17 mm/well) and acetone (16 mm/well) extracts. The widest inhibition zone of 23 mm/well in diameter was obtained from Aghormy cyanobacterial mats extracted in n-hexane against S. aureus. Non-polar extracts had non-significant highest antibacterial activities at both concentrations of 1 and 5 mg mL–1 at different lakes, except at Aghormy Lake at concentration of 1 mg mL–1 where the polar extracts were more effective against the tested microbs.
Chemical composition of the extracts: Chemical composition of extracts of cyanobacterial mats was compared using GC-MS. A total of 44 chemical compounds were found from all mats (Table 2). The chemical composition of cyanobacterial mats varied from one lake to the other with the highest number of compounds found in Zieton mats (38 compounds), whereas the lowest number was found in Aghormy mats (31 compounds). The highest number of compound (11 compounds) was extracted by ethyl acetate in Aghormy Lake, by methanol in Zieton Lake and acetone in Maraqi Lake. No chemical compounds were detected by methanol and ethanol from Aghormy and Zieton lakes, respectively. The chemical profiles of cyanobacterial mat extracts from Aghormy, Zieton and Maraqi mats shared similarity more than 40% with highest similarity (52.53%) between Aghormy and Maraqi extracts (Fig. 2).
Between the detected metabolites, 25 compounds were unique and were present only in extract of particular cyanobacterial mat but absent in the others. Some compounds were unique for cyanobacterial mat of specific lake but were not present in the other lakes.
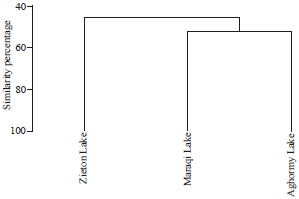 | |
| Fig. 2: | Cluster analysis of similarity between GC-MS chromatograms of polar and non-polar extracts from the three studied cyanobacterial mats |
Twelve unique compounds were extracted from Zieton cyanobacterial mat, seven unique compounds were extracted from Aghormy cyanobacterial mat and six unique compounds were extracted from Maraqi cyanobacterial mat. Fourteen unique compounds were extracted by the polar solvents, whereas eleven unique compounds were extracted by the non-polar solvents. The highest number of compounds were extracted by ethyl acetate (27 compounds) followed by acetone (26 compounds). All lakes extract contained a suite of fatty acids, amino acids, alcohols, esters and benzene derivatives.
Phytochemical analysis: The results of phytochemical screening of extracts revealed the presence of terpenoids, alkaloids, saponins and glycosides in the cyanobacterial mat of the three lakes (Table 3). Both Phenolic compounds and flavonoids were completely absent from the cyanobacterial mat in the three lakes. Lake Zieton is a good source of both terpenoids and glycosides compounds, where all polar and non-polar solvent are effective to isolate these compounds. The non-polar solvent, particularly petroleum ether and n-hexane, are the most effective for isolate active saponins, especially in Aghormy and Zieton Lakes.
The bioactivity of mixed cyanobacterial and aerobic heterotrophic bacterial community forming the top mat layer was tested. These cyanobacteria-dominant mat layers were shown to exhibit in vivo antibacterial inhibitory activities. This approach takes advantage of in situ cell to cell relationship in multispecies communities that does not exist in pure cultures, as well as the isolation of compounds that might be produced by uncultured microorganisms (Dobretsov et al., 2011).
| Table 2: | Chemical composition of cyanobacterial mats' extracts as revealed by gas chromatography mass spectrometry (GC-MS) |
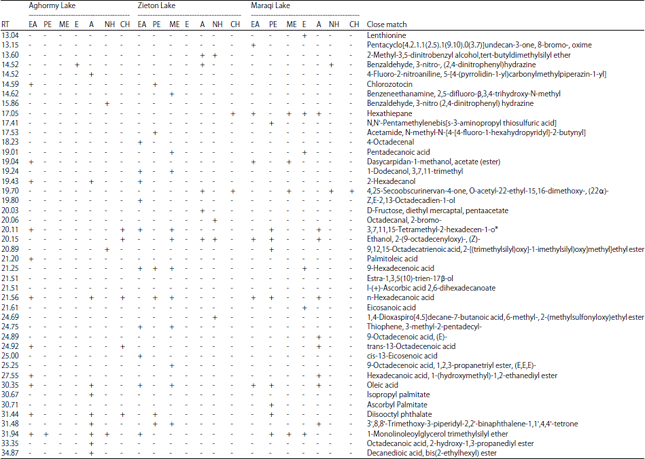 | |
| RT: Retention time (min), EA: Ethyl acetate, PE: Petroleum ether, ME: Methanol, E: Ethanol, A: Acetone, NH: n-Hexane and CH: Chloroform | |
| Table 3: | Phytochemical composition of cyanobacterial mat extracts |
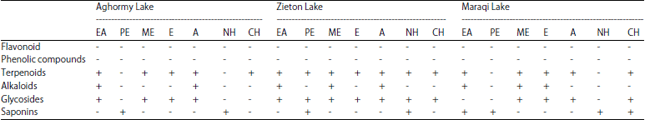 | |
| EA: Ethyl acetate, PE: Petroleum ether, ME: Methanol, E: Ethanol, A: Acetone, NH: n-hexane and CH: Chloroform | |
Pseudomonas aeruginosa and C. albicans showed no inhibition at lower concentrations of 1 mg mL–1 of different extracts, whereas S. aureus and Aeromonas sp. were the most susceptible bacteria at the same concentration. Abed et al. (2013) examined the effect of 1 mg mL–1 extracts of cyanobacterial strains isolated from hyper saline pools and cultured in hyper saline synthetic media against 9 pathogens. They found that P. aeruginosa and C. albicans were the most resistant and showed no inhibition, whereas S. aureus was one of the most susceptible pathogens. They conducted that microorganisms from these mats are not exposed to pathogens and thus, do not produce antibiotics against them. The results of this study indicated that higher concentrations of 5 mg mL–1 can effectively inhibited all pathogens except P. aeruginosa, which indicated the fact that cyanobacterial mats of Siwa lakes produce diverse antibiotic compounds although they are not exposed to pathogens but may be at low concentrations. Many reports indicated that P. aeruginosa and C. albicans, one/both, showed high resistance against cyanobacterial extracts (Issa, 1999; Khairy and El-Kassas, 2010) and plant extract (Hossain et al., 2014; Mohamed et al., 2014; Barreca et al., 2014). Giamarellos-Bourboulis et al. (1999) and Khanna and Kannabiran (2008) indicated that P. aeruginosa is a multi-resistant pathogen and due to lack of active antibiotics against this bacterium increasing incidence of nosocomial infections and high mortality. As estimated, cyanobacterial mat extracts activity against Gram -ve, except P. aeruginosa and the tested Gram +ve bacteria and the tested yeasts was established from this study. This may be attributed to divers active compounds present in cyanobacterial extracts as reported by previous studies (Ozdemir et al., 2004; Khairy and El-Kassas, 2010).
Non-polar extracts had a significant higher antibacterial and antifungal activities against the tested microbes compared with the polar extracts. Whereas, polar extracts of Aghormy cyanobacterial mat had, also, non-significant higher antimicrobial activities. Previous studies performed on extracts of cyanobacterial isolates from microbial mats (Biondi et al., 2008), extracts of cyanobacterial mat from hot springs (Dobretsov et al., 2011) and extracts of cyanobacterial mat from hyper saline stream (Abed et al., 2011), also extracts of Anabaena flos aquae (Khairy and El-Kassas, 2010) conducted that the non-polar extract of ethyl acetate exhibited the highest antibacterial, antifungal and cytotoxic activities. Many other studies showed that the polar extracts of methanol are more effective than non-polar extracts of ethyl acetate (Hossain et al., 2014; Mohamed et al., 2014).
The strong antimicrobial activity of the different extracts was attributed to the abundance of many compound characterizes by antifungal and antibacterial activities. The GC-MS analysis of different extracts of cyanobacterial mats revealed high abundance of saturated and unsaturated fatty acids, their esters, polyalcohols, benzene derivatives and many other compounds (Table 1 GC-mass). Many fatty acids were recorded as antimicrobial agents (Wu et al., 2006; El Semary, 2012a). They reported that some fatty acids have cytotoxic effects on other organisms. The free fatty acids were also reported to be potent allelopathic agents (Ramsewak et al., 2001). Wu et al. (2006) attributed the cytotoxicity effects of fatty acids to their ability to increase the membrane permeability leading to membrane damage. Mundt et al. (2003) suggested that fatty acids produced by cyanobacteria as a defense mechanism against other microorganisms might be able to change the permeability of the cell membrane through interacting with proteins and lipids of the membrane, inhibiting special enzymes or by forming a layer around the cells. Some fatty acid, oleic acid, exhibits a considerable activity against some micro-organisms (Novak et al., 1961). Quartz crystal microbalance with dissipation data indicated an essentially non-disruptive binding of oleic acid to supported lipid bilayers, leading to formation of highly dissipative and "soft" lipid membrane (Nielsen et al., 2010). Other fatty acids, palmitoleic acid, are associated with a concerted reduction in the fatty acid synthase II activity with respect to the control lines and an increase of stearoyl-ACP desaturase activity (Salas et al., 2004). Many studies have shown that compounds of benzene derivatives not only exhibited antibacterial activities (Cushnie and Lamb, 2005; Lee et al., 2009), but also the antibacterial activity of some benzene containing compounds is superior to that of benzene containing drugs (Bankova et al., 1996). It has previously been shown that some benzene inhibited b-ketoacyl-acyl carrier protein synthase III, a condensing enzyme that initiates fatty acid biosynthesis in most bacteria, leading to antimicrobial activity (Lee et al., 2009).
The phytochemical screening revealed the presence of terpenoids, alkaloids, saponins and glycosides and the absence of Phenolic compounds and flavonoids. Some of these substances were detected in cyanobacterial extracts and were reported to have antimicrobial effects (Kulik, 1995; El Semary, 2012b). Terpenoids and glycosides were the highly detected compounds by most solvents in the three lakes which revealed that these lakes may be rich by these compounds. Terpenoids such as triterpenes, sesquiterpenes and diterpenes have been referred to as antibiotics, insecticidal, anthelmintic and antiseptic in pharmaceutical industry (Parveen et al., 2010). The terpenoid fraction had weak antimicrobial activity against P. aeruginosa and E. coli (Feio et al., 2002; Mastelic et al., 2005) but cause high growth reductions of the medically important pathogen S. aureus and C. albicans, both were inhibited at a minimal concentration of 5 mg mL–1 (Mastelic et al., 2005). Matsumura et al. (1990) and Bilkova et al. (2015) studied the effect of different length chain glycosides on different pathogens and found that S. aureus and C. albicans were the most susceptible pathogens and showed potent activity at micromolar level, whereas E. coli was the least affected microorganism by the tested compounds. Many reports indicated that natural alkaloids (Singh and Sharma, 2013; Hu et al., 2014) and natural saponins (Khanna and Kannabiran, 2008; Wang et al., 2012) are highly effective against a wide spectrum of pathogens.
The results indicated that both polar and non-polar solvents are effective to extract cyanobacterial mats from northern western desert. The tested pathogens were highly susceptible to all extracts except P. aeruginosa especially at highe concentrations. The GC-MS showed that the extracts of the cyanobacterial mats contains divers' compounds specifically fatty acids. Also, the cyanobacterial mats are rich with the high molecular weight compound; terpenoids, alkaloids, saponins and glycosides.
The author is very grateful to Prof. Shawky Sabea, head of Fresh water and Lakes Division, National Institute of Fisheries and Aquatic Science for his continuous support during the work. The author acknowledged Dr. Manal El Barbery and Mr. Ehab Shawer for their providing the microbial cultures for the experiments and sincere gratitude for Mr. Ehab Shawer who carried out the antimicrobial assays.