Research Article
Application of Geoaccumulation Index and Enrichment Factors on the Assessment of Heavy Metal Pollution along Irbid/zarqa Highway-Jordan
Department of Earth and Environmental Sciences, Faculty of Science, Yarmouk University, Jordan
Heavy metals are natural constituents of sediments. They come from rocks and soils through the processes of erosion, transport and deposition. They can also be derived from anthropogenic sources in which case they are incorporated into sediments as artificial pollutants from industrial or urban releases and wastes (Benamar et al., 1999).
Road dust originates from the interaction of solid, liquid and gaseous materials which are produced from different sources and deposited on a road. The composition and quantity of chemical matrix of road dust are indicators of environmental pollution (Banerjee, 2003). Road dust receives varying inputs of heavy metals from diversity of mobile or stationary sources such as vehicular emission, industrial plants, power generation plants, oil burning, waste incineration, construction and demolition activities as well as resuspension of surrounding contaminated soils (Ahmed and Ishiga, 2006; Al-Khashman, 2007). Lead (Pb), for example is known to come from the use of leaded gasoline whereas Cu, Zn and Cd from tyre abrasion, lubricants, industrial and incinerator emissions (Thorpe and Harrison, 2008; Wilcke and Zech, 1998). The source of Ni and Cr in street dust is believed to be due to corrosion of vehicular parts Lu et al. (2009), Akhter and Madany (1993) and Fergusson (1991) and chrome plating of some motor vehicle parts (Al-Shayep and Seaward, 2001). The phenomenon contributes significantly to the pollution of urban environment. This makes the study of sediments important for determining the origin, distribution and level of heavy metal in urban surface environments.
However, the quantitative data on heavy metal concentrations, their contamination levels and their pollution sources have not been systematically gathered and intercompared. Therefore, this study focuses on heavy metal contamination in urban soils. The sources, concentrations, pollution levels, sample collection and analytical tools of heavy metals are elucidated in this study.
Study area: The study area, situated in the northern part of Jordan limited between 35°75’-36°10’N and between 32°25’-32°45’E (Fig. 1).
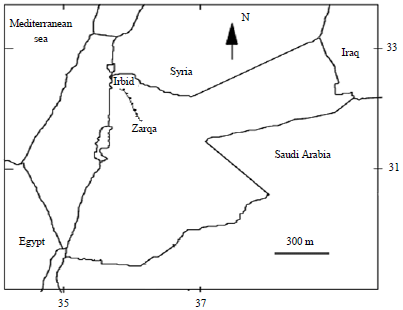 | |
| Fig. 1: | Location map of the study area |
The climate of the study area is Mediterranean with more than 370 mm mean annual rainfall that is decreasing gradually from the west (450 mm year–1) to the east (200 mm year–1). Mean winter air temperature ranges from 5-9°C and mean summer air temperature ranges from 22-29°C (Khresat et al., 1998). The investigated area is dominated by Amman Silicified Limestone Formation from the Companian age. This unit consists of limestone, chert, massive chalky and marl (Chen et al., 2007). Large parts of the studied area are covered by soil. The soil in the investigated area is yellow-brown with calcareous concretions. It is composed of loess-like silt and residual calcareous bedrock.
Sediments sampling: Twenty one soil samples were collected during January 2011 from different depth with an interval of 10-30 cm. The 1 kg of each soil sample were collected using a stainless steel spade and a plastic scoop, All samples collected were stored in sealed polythene bags and transported to the laboratory for pre-treatment and analyses.
Chemical analysis: The soil samples were dried, mechanically in the laboratory, the soil samples after air drying at room temperature, were sieved with nylon mesh (2 mm). The <2 mm fraction was grinded in an agate and pestle and passed through a 63 micron sieve. Soil samples were analyzed for heavy metals. These samples were digested in aqua regia (1:3 HNO3: HCL). Metals in the final solutions were determined using Pyeunicam, SPQ PHilips, Atomic Absorption Spectrometer (AAS). Standard stock solutions for all the elements were procured from Merck as well as prepared in the laboratory following the procedures as described in APHA (1989). The glassware used was Pyrex, which was washed several times with soap, distilled water and diluted nitric acid to remove any impurities.
Contamination assessment methods: The assessment of soil or sediment enrichment can be carried out in many ways. The most common ones are the index of geoaccumulation and enrichment factors (Lu et al., 2009). In this work, the index of geoaccumulation (Igeo) and Enrichment Factor (EF) have been applied to assess heavy metals (As, Cr, Cu, Mn, Ni, Pb and Zn) distribution and contamination in the road dust samples along Irbid-Zarqa highway.
A quantitative measure of the extent of metal pollution in the studied soil was calculated using the geoaccumulation index proposed by Muller (1969). This index (Igeo) of heavy metal is calculated by computing the base 2 logarithm of the measured total concentration of the metal over its background concentration using the following mathematical relation (Ntekim et al., 1993):
Igeo = log2 (Cn/1.5Bn)
where, Cn is the measured total concentration of the element n in the mud grain size fraction of sediment, Bn is the average (crustal) concentration of element n in shale (background) and 1.5 is the factor compensating the background data (correction factor) due to lithogenic effects. Loska et al. (2003) gave the following interpretation for the geoaccumulation index: Igeo<0 = practically unpolluted, 0<Igeo<1 = unpolluted to moderated polluted, 1<Igeo<2 = moderately polluted, 2<Igeo<3 = moderately to strongly polluted, 3<Igeo<4=strongly polluted, 4< Igeo<5= strongly to extremely polluted and Igeo >5 = extremely polluted.
Enrichment factor: Enrichment Factor (EF) has been employed for the assessment of contamination in various environmental media by several researchers. Its version adapted to assess the contamination of various environmental media is as follows:
EF = (Cx/Cref)Sample
(Bx/Bref)Background
| Cx | = | Content of the examined element in the examined environment |
| Cref | = | Content of the examined element in the reference environment |
| Bx | = | Content of the reference element in the examined environment |
| Bref | = | Content of the reference element in the reference environment |
An element is regarded as a reference element if it is of low occurrence variability and is present in the element in trace amounts. It is also possible to apply an element of geochemical nature whose substantial amounts occur in the environment but has no characteristic effects i.e. synergism or antagonism towards an examined element. The contamination categories are recognized on the basis of the enrichment factor: EF<2 states deficiency to minimal enrichment, EF = 2-5 moderate enrichment, EF = 5-20 severe enrichment, EF = 20-40 very high enrichment and EF>40 extremely high enrichment (Manno et al., 2006). "Metal distribution in road dust samples collected in an urban area close to a petrochemical plant at Gela, Sicily", Atmos Environ, 40, 5929-5941, Al-Khashman (2007). Despite certain short comings, Aikpokpodion et al. (2010). The enrichment factor, due to its universal formula is relatively simple and easy tool for assessing enrichment degree and comparing the contamination of different environment.
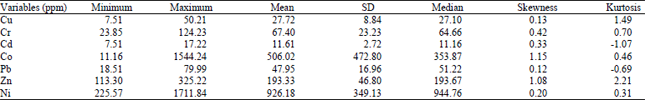
Heavy metal concentrations: Table 1 summarizes the minimum, maximum, mean, standard deviation and median concentrations of a number of metals (Zn, Cr, Co, Pb, Ni, Cu and Cd) in twenty one soil samples collected along Irbid/Zarqa highway of Jordan. A close look at Table 1 shows that the variability in the range of all the metal distributions as compared with their means respectively is an indication of pollution of the sample with that metal ion. The decreasing trend of averages of metal levels was as follows: Ni>Co>Zn >Cr>Pb>Cu>Cd.
Distribution and enrichment of metals: The Enrichment Factor (EF) of Co, Cr, Ni and Cu concentration in the soil are 5.33, 0.75, 12.35, 0.40, respectively (Table 2) and the calculated results of Igeo for these heavy metals have Igeo≤0 indicating that the environment is unpolluted by these elements.
Meanwhile, Enrichment Factor (EF) of Pb and Zn recorded 2.40 and 2.04, whereas, the value of the geoaccumulation index are 0.21 and 0.13, respectively which indicates that pb and Zn exhibits from unpolluted to moderately polluted. The soils of the study area were mainly derived from the upper cretaceous carbonate rocks. But carbonates in general have low concentrations of lead. It is clear that the carbonate parent rocks are not the only source of lead and it must be other sources which are more likely anthropogenic ones. Since the soil samples have been taken along highways with considrable heavy traffic rates, so the motor vehicles burning leaded gasoline can be considered as the main source of the lead in the soils of the study area.
The behavior of Cd show that the Enrichment Factor (EF) is 38.7, whereas, the value of the geoaccumulation index is 1.41. Relatively higher values of cadimium concentrations in the analyzed soil samples reflect anthropogenic effects may be burning of fossil fuel, wear and tear of tyres (Kabata-Pendias and Pendias, 1984). The soil lying at the vicinity of high ways may have a cadmium ranging from 1-10 ppm. This may explain the relatively high concentrations of Cd in the soils of the study area.
| Table 1: | Basic statistical parameters for the distribution of heavy metals in the investigated soil samples |
 | |
| Table 2: | Average, background, enrichment factor, calculated I-geo index and grade of pollution intensity of heavy metals in metals in soil samples |
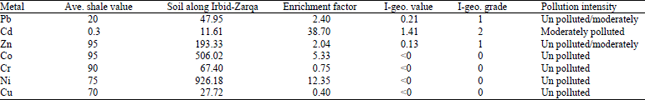 | |
This study revealed that the soil along Irbid-Zarqa highway during January 2011 is predominantly by Cd>Ni>Co>pb>Zn>Cr>Cu. As recorded the highest EF value at 38.7 followed by Ni (12.35), Co (5.33), Pb (2.40), Zn (2.04), Cr (0.75) and Cu (0.40). And the mean Igeo provided the same trend of pollution levels as in the case of the EF, which indicates that the highest level goes to Cd (1.41), exhibits moderately polluted. Meanwhile, Pb recorded 0.20 and Zn (0.13), which illustrates that both of these elements vary from unpolluted to moderately polluted. The Co, Cr, Ni and Cu levels are below 0, which demonstrates background concentrations. The distribution of metal concentration in the study area has come about as a result of anthropogenic influences, in particular vehicular emissions.