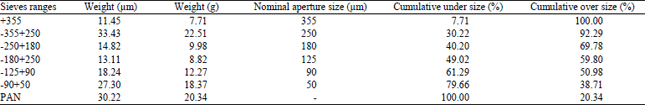
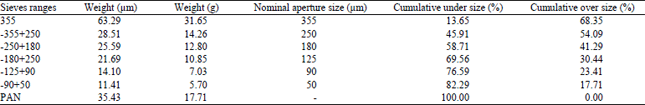
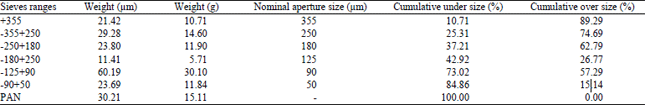
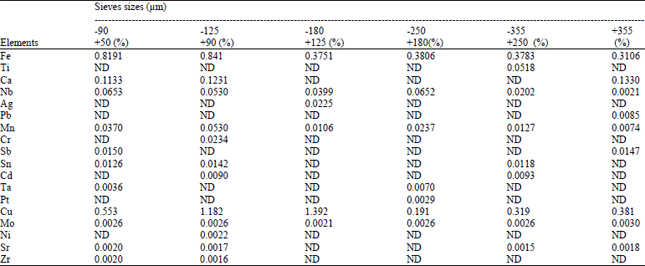
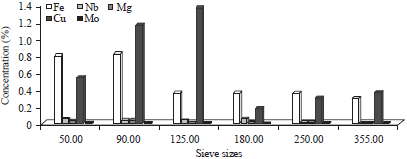
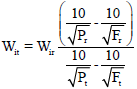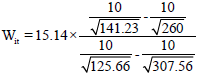Research Article
Physiochemical Analysis of Chanchaga Ore, North Central Nigeria
Department of Mechanical Engineering, Federal University of Technology, Minna, Nigeria
A.S. Abdulrahman
Department of Mechanical Engineering, Federal University of Technology, Minna, Nigeria