Research Article
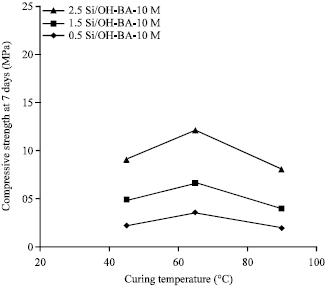
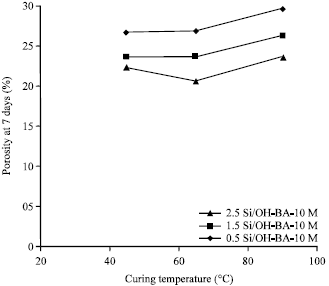
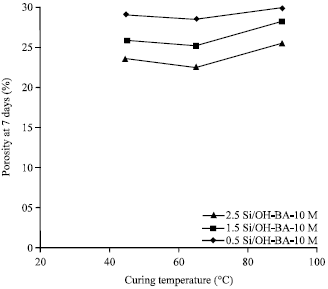
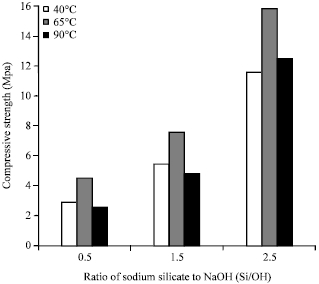
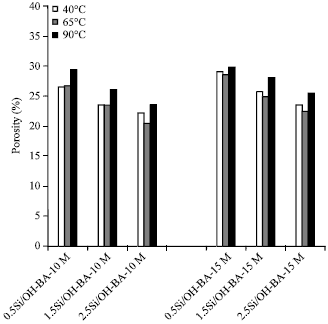
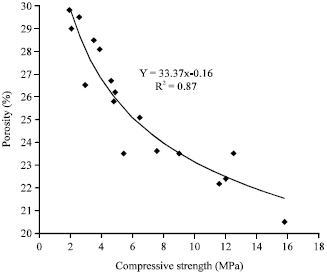
Strength and Porosity of Bagasse Ash-based Geopolymer Mortar
Department of Civil Engineering, Rajamangala University of Technology Phra Nakhon, Bangkok, 10300, Thailand
Prinya Chindaprasirt
Sustainable Infrastructure Research and Development Center, Department of Civil Engineering, Faculty of Engineering, Khon Kaen University, Khon Kaen, 40002, Thailand