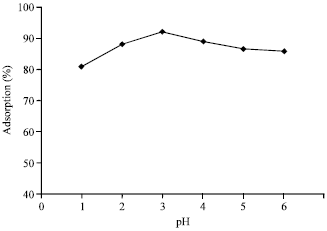
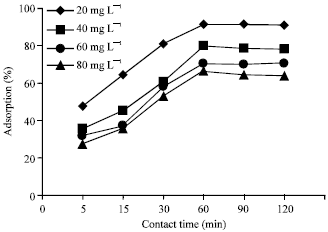
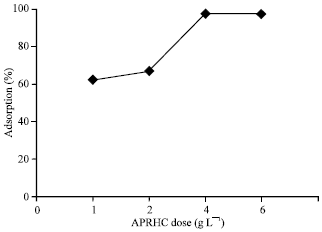
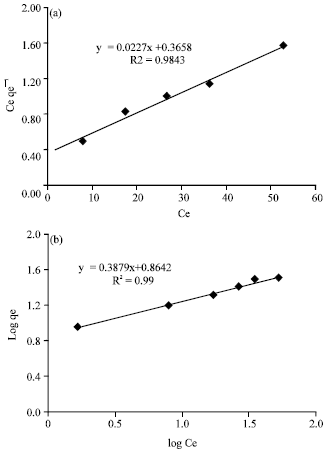
Research Article
Acidically Prepared Rice Husk Carbon for Adsorption of Zn(II) from Aqueous Solution
Department of Civil Engineering, Universiti Teknologi PETRONAS, Perak Darul Ridzuan, 31750, Tronoh, Malaysia
Mohamed Hasnain Isa
Department of Civil Engineering, Universiti Teknologi PETRONAS, Perak Darul Ridzuan, 31750, Tronoh, Malaysia
Malay Chaudhuri
Department of Civil Engineering, Universiti Teknologi PETRONAS, Perak Darul Ridzuan, 31750, Tronoh, Malaysia
Muhammad Raza Ul Mustafa
Department of Civil Engineering, Universiti Teknologi PETRONAS, Perak Darul Ridzuan, 31750, Tronoh, Malaysia