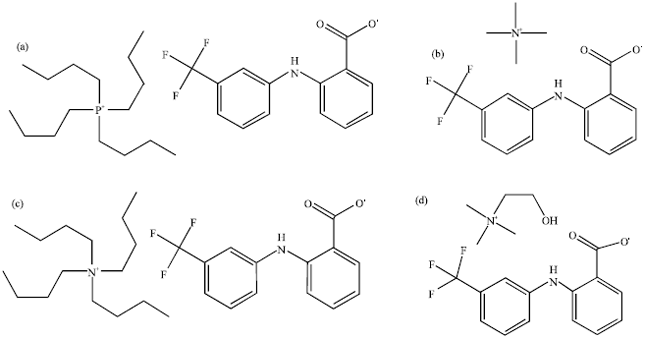
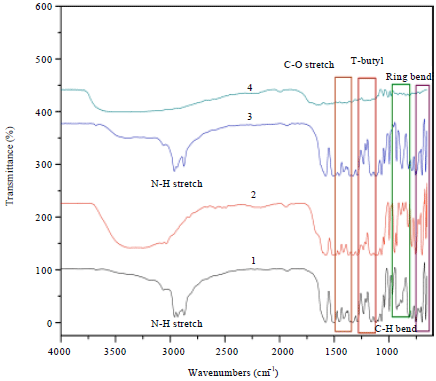
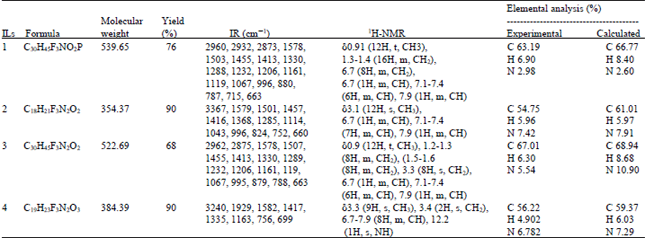
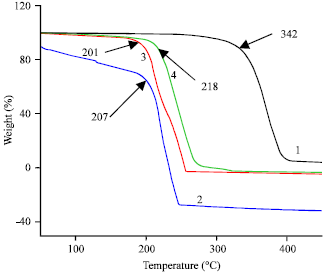
Research Article
Synthesis and Characterization of Flufenamic Ionic Liquids
Department of Chemical Engineering, 37150, Tronoh Perak, Malaysia
Cecilia Devi Wilfred
Department of Fundamental and Applied Science, Universiti Teknologi, PETRONAS, 37150, Tronoh Perak, Malaysia