Research Article
Effectiveness of Local Exhaust Ventilation Systems in Reducing Personal Exposure
Petronas Chemicals Group Berhad, Level 21, Tower 2, PETRONAS Twin Towers, Kuala Lumpur, Malaysia
This study attempts to develop a workplace protection factor for LEVs. The workplace protection factor (WPF) of LEV is defined as providing the minimum degree of protection from hazardous substances exposure in the workplace. The LEV protection factor figure can be used in the Cherrie model to estimate the past exposure level (Cherrie and Schneider, 1999). Therefore, the protection factor can be used as a guidance to estimate the worst case and the best case scenarios in order to estimate exposure levels for at least similar process in similar industry.
Most literatures focused on the workplace/assigned protection factors for respirators. The WPF is calculated by dividing the ambient concentration by the concentration measured inside the respirator (Myers and Zhuang, 1998). Following the same logic, the WPF for LEV is calculated by dividing the concentration when LEV is off by the concentration when LEV is on. WPF data collected from field studies, researchers and research organizations were evaluated by the ANSI committee who set the Assigned Protection Factor (APF) for respiratory protection (Nelson, 1996).
The hazard ratio is defined by ANSI as the measured or estimated exposure in the workplace divided by the Occupational Exposure Limit (OEL) (Nelson, 1996; Nelson et al., 2000). The APF is very important in ensuring the selection of a respirator which is sufficient to overcome the expected concentrations at the workplace (Nelson, 1996).
At the workplace, the Maximum Use Concentration (MUC) is also important when selecting the respirator to be used in a given setting. The MUC is the concentration of the OEL multiplied by the APF (Nelson et al., 2000). If a respirator is required at the workplace, the OEL multiplied by the APF must be less than the Maximum Use Concentration (MUC) (Nelson et al., 2000). For example, an APF half-face piece is 10, the MUC is 10 times the exposure limit (Nelson et al., 2000).
Industries studied: The LEV systems were selected based on the possibility to conduct face velocity measurement to determine their effectiveness. Two different solvents and particulates were measured for the different types of LEV systems. Five types of working environment were involved in this study: a chemistry laboratory, a Motor Vehicle Repair (MVR) workshop, a painting mixing facility, a wood cutting factory and a construction/fabrication plant.
Measurement of face velocity: The face velocity is the air velocity at the hood opening (ACGIH, 1998). The face velocity is one of the methods to determine the effectiveness of LEV systems which is based on maintenance, examination and testing of the local exhaust ventilation HSG 54 and the laboratory fume cupboard (BSI, 1994). It will determine the effectiveness of LEV systems if there are no changes as per designed (Maupins and Hitchings, 1998 and Diberardinis et al., 2003). The hot-wire anemometer was used to measure the face velocity for this research. All face velocity measurements taken did not deviate by more than ± 20% from the averages face velocity (BS 7258: Part 1, BSI, 1994).
Fume hood cupboard face velocity measurement: All apparatus inside the fume hood need to be removed for accurate face velocity measurement (Maupins and Hitchings, 1998). The sash was opened 500 mm in the normal working position and the baffle was set to the normal position. The face velocity is normally specified in accordance with a sash working height of 500 mm (BSI, 2003). The sash opening height was divided by two peripheral rectangles.
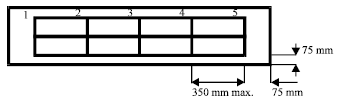 | |
| Fig. 1: | Hood opening for face velocity measurement |
The width of the peripheral rectangular did not exceed a distance of 350 and 75 mm (Fig. 1) from the sides of the opening (BS 7258: Part 1, BSI, 1994). The peripheral rectangles were crisscrossed with a number of thin cotton strings. Face velocity readings were taken with a hot wire anemometer placed at position 1, 2 and others as shown in Fig. 1 (BS 7258: Part 1, BSI, 1994). The anemometer was held horizontally in the plane of the sash opening for 15 sec to allow the face velocity to stabilize. The readings were taken three times at each point. The averages of the face velocity were calculated from the measurements (BS 7258: Part 1, BSI, 1994). When measuring, care was taken to stand at the fume hood corner so as not to affect the airflow. To avoid air draughts, the laboratory door was also closed during face velocity measurement (Maupins and Hitchings, 1998).
Spray booth face velocity measurement: All equipment needs to be removed from the spray booth to ensure accuracy of the face velocity measurement. Doors were closed during face velocity measurement to avoid air draught as well as to prevent air supply from moving out of the spray booth (HSE, 2000a). The area of down-draft filter opening was identified by the owner to ensure the face velocity was taken at the down-draft hood. The down-draft filter opening was divided by equal area rectangles (350x350 mm) as shown in Fig. 2.
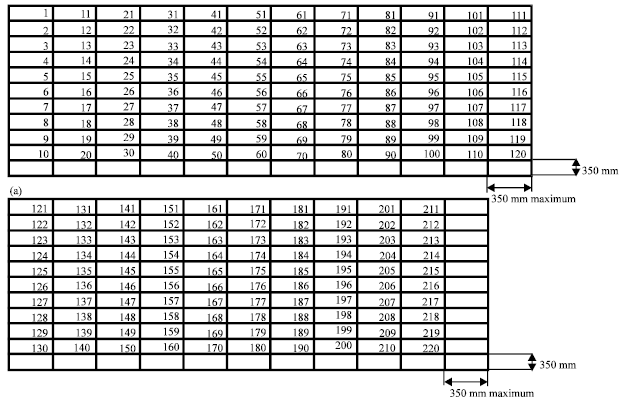 | |
| Fig. 2: | Face velocity measurement of spray booth, A: Continued and mm: Millimeter |
The peripheral rectangles were crisscrossed with a number of thin cotton strings. Face velocity readings were taken with a hot wire anemometer placed at position 1, 2 and others as Fig. 1 (BSI, 1994). The anemometer was held horizontally in the plane of the down-draft filter opening for 15 seconds to allow the face velocity to stabilize. The readings were taken three times at each point. When measuring, care was taken to stand away from the down-draft filter opening so as not to affect the airflow. The averages of the face velocity were calculated from the measurements (BSI, 1994).
Movable or partial paint mixing hoods face velocity measurement: The movable paint and partial hoods diameters were 0.15 and 0.1 m, respectively. The hood exterior was visually inspected to ensure that no movable hoses were damaged and connection to the ducting was maintained in good condition. Door opening was avoided and that no other movable hoods were working nearby during face velocity measurement to avoid any disturbance. Face velocity was measured at the center of the opening. The anemometer was held horizontally in the plane of the opening for a few seconds to allow the face velocity to stabilize. The readings were taken three times at the central opening. The averages of the face velocity were calculated from the measurements (Maupins and Hitchings, 1998).
Radial arm saw face velocity measurement: The diameter of the radial arm saw LEV system hood piping was 0.1 m. Before the face velocity measurement was taken, the radial arm saw was on for about 5-10 min to ensure that there was no dust accumulated inside the ducting. Face velocity was measured at the center of the opening. The anemometer was held horizontally in the plane of the opening for a few seconds to allow the face velocity to stabilize. The readings were taken three times at the central opening. The averages of the face velocity were calculated from the measurements (Maupins and Hitchings, 1998).
Movable welding hood face velocity measurement: The diameter of the movable welding hood was 0.3 m. A visual inspection of the LEV system was done to ensure that there is no damage to the flexible hose and that the connection to the ducting was in good condition. Door opening was avoided as well as ensuring that no other movable welding hood is working nearby during face velocity measurement to avoid any disturbance. Face velocity was measured at the centre of the opening. The anemometer was held horizontally in the plane of the opening for a few seconds to allow the face velocity to stabilize. The readings were taken three times at the central opening. The arithmetic mean velocity was calculated from these measurements (Maupins and Hitchings, 1998).
Personal monitoring overview: Personal monitoring is the measurement of a particular employee’s exposure to airborne contaminants that reflects actual exposure to the employee (Nims, 1999). The personal monitoring method was based on the Health and Safety Executive (HSE) Methods for the Determination of Hazardous Substances (MDHS) 14/3 and 70 for Respirable and Inhalable Dust and Gases and Vapors respectively (HSE, 2000b and HSE, 1994). The solvent samples were analyzed by a laboratory chemist of the Institute of Occupational Medicine (IOM) refers to the HSE under Methods for the Determination of Hazardous Substances (MDHS) 96 for Volatile Organic Compounds in Air (4) (HSE, 2000c). The particulate samples were analyzed by the author at the laboratory of the Department of Environmental and Occupational Medicine using gravimetric analysis as explained in 4.3.7 (HSE, 2000a).
Personal monitoring for diethyl ether at chemistry laboratory: A calibrated pump (SKC, Incorporation) and coconut shell charcoal tubes (SKC 226-01) was used to collect the diethyl ether vapour. The charcoal tube was broken at each end, inserted into the adapter tube and the other end of the tube connected to the pump (HSE, 1994). The charcoal tube position followed the arrow as indicated at the charcoal tube for air flow intake to the pump. The pump flow rate was set at 0.2 l min-1 using a rotameter. A low flow before sampling and calibration data was recorded on each occasion. The sampling head was attached to the worker’s upper chest, not more than 30 cm away from the nose-mouth region (HSE, 2000b). The duration of the monitoring was approximately 15 min for task. Three samples were taken with the LEV system switched off and later, with the ventilation system switched on. The fume hood cupboard was on for about 5-10 min after each sampling to minimize the contaminant inside the fume hood cupboard. At the end of the sampling period, the pump flow rate was also rechecked and the value noted (HSE, 1994; Ashton and Gill, 1992). The pump was then stopped and the time noted. The charcoal tube was sealed with plastic caps at each end of the tube, labeled properly and immediately stored in a 4°C fridge.
A field blank was brought in for the sampling process. It was opened to the workplace environment during the charcoal tube preparation for sampling. Two of the total number of diethyl ether samples were field blanks. All the sampling tubes were sent to the IOM laboratory for analysis of the diethyl ether mass and the concentration was calculated as described in Eq. 1.
| (1) |
Personal monitoring for xylene at paint facilities: Sampling for xylene was carried out at the MVR workshops. The method employed was identical to that described earlier. The duration of the monitoring of the spray booths was 5 min for each sampling. Three samples were taken during on and off ventilation periods respectively. In spray booths 2 and 3, the spray gun was located at the end of a down-draft filter by using a stand at a normal working position height instead of a worker holding the spray gun. The pump and tube holder were placed near to the spray gun, as close as possible to the normal working position. This was due to the company procedure that does not allow anybody to be in the spray booth when the LEV system is switched off. The sprayer was adjusted to produce a constant flow rate. In spray booth 1, the spray gun was held and the tube holder was attached to the sprayer’s collar. The pump was placed in his coverall pocket. At the end of the sampling period, the pump was stopped and the time noted. The pump flow rate was also rechecked and the value noted (HSE, 1994; Ashton and Gill, 1992). The spray booth was on for about 5-10 min after each sample was taken to minimize the contaminant inside the spray booth. The charcoal tube was sealed with plastic caps at each end of the tube, labeled properly and immediately stored in a 4°C fridge.
A field blank was brought in for the sampling process. It was opened to the workplace environment during the charcoal tube preparation for sampling. Two of the total numbers of xylene samples were field blanks. All the sampling tubes were sent to the IOM laboratory to produce the xylene mass and the concentration as calculated in Eq. 2:
| (2) |
Personal monitoring for xylene at movable and partial paint hoods: Sampling for xylene was also carried out at the paint manufacturing plant. The method employed was similar to that described in section 2.3.2. Due to the higher xylene volumes employed, the pump was set at a lower sampling rate of 0.1 l/min. The duration of the monitoring of the movable and partial paint hoods was approximately 15 min for each sampling. The tube holder and pump were placed near to the pot tank opening at the normal working position height. Field blanks were collected as described previously.
Personal monitoring for total inhalable dust at the wood cutting plant: Personal sampling for total inhalable dust was carried out in accordance with MDHS 14/3 (HSE, 2000c). A calibrated pump (SKC, Incorporation) with a seven hole sampling head was used to collect the wood dust. All collections used glass microfibre filters (25 mm diameter). The filters were kept in the sample cans to transport to the site. The pump was switched on for a few min before calibration to stabilize the pump (HSE, 2000a). The pump flow-rate was calibrated at 2.0 l min-1 using a rotameter. The sampling head was attached to the worker’s upper chest, not more than 30 cm away from the breathing zone (HSE, 2000b). The pump was placed in the worker’s coverall pocket or hung on his belt. The timer was started and the time noted when cutting began. At the end of the sampling period, the pump was stopped and the time was taken. The pump flow rate was checked and noted at the end of sampling. The filter was removed with forceps and placed in a labeled container. Every sampling was taken six times each, for 15 min. Three samples were taken during the on and off ventilation periods, respectively.
The field blank was brought in during the sampling. The field blank was one taken from each batch of ten prepared (HSE, 2000c). All filters were sent to the laboratory for gravimetric analysis.
Personal monitoring for welding fume: Sampling for welding fume was carried out in accordance with MDHS 14/3 (HSE, 2000a). The procedure was identical to that described in 2.3.5 with the exception that the sampling head was attached to an aluminium steel plate and was hung from the headband inside the worker’s visor (HSE, 1990).
Gravimetric analysis: The glass microfibre filters were pre conditioned before and after sampling by placing them in open sample cans overnight in the weighing room (the weight of filters tends to vary with the humidity) (HSE, 1990). All samples were weighed before and after sampling in the laboratory room. All filters were handled with forceps. The initial weight and pre-conditioned weight filter readings were taken to produce the average of initial filter weight. The particulates concentration was calculated as Eq. 3.
| (3) |
Monte Carlo simulation: The exposure concentration results from the workplace personal monitoring were used to produce the LEV Workplace Protection Factor (WPF). The average of personal exposure concentration with the LEV switched off was divided by the average of exposure concentration when the LEV was switched on as in Eq. 4:
| (4) |
The WPF results were analyzed using the Statistical Package for Social Sciences (SPSS). All measurement results were transformed to log - normal distribution. The WPF Geometric Mean (GM) and the Geometric Standard Deviation (GSD) from SPSS were entered as input for the Monte Carlo (MC) simulation. Other variables also were determined as input for the MC simulation. The Cherrie model (Cherrie and Schneider, 1999) was used to determine the exposure level using information on the exposure determinants.
Based on the work done by Cherrie and Schneider (1999) three parameters were used: method of handling, LEV (WPF) and dilution or general ventilation (air changes per hour). All these parameters were multiplied to produce the exposure concentration. 10,000 repeated MC simulations calculated each variable in order to produce distribution of exposures concentration outcomes. MC simulation will determine the uncertainty of variables in the model. Finally, the sensitivity analysis showed the highest value of variables that contribute to exposure concentration.
The inputs that were entered into the MC simulation for the fume cupboard LEV systems. The inputs included the quantity of diethyl ether used, LEV WPF and the air changes per hour in the laboratory (CIBSE, 2001). The inputs included the quantity of xylene used in the spray booth, the spray gun opening for spraying, LEV and air changes per hour.
All these figures were entered into the MC simulation relative to unity to determine the contribution to total exposure from each parameter. All variable values were divided by the most likely and geometric or arithmetic means to produce a unity.
In the @RISK software (Risk 4.0, 2000), three distribution functions were used: RiskTriang, RiskLognorm and RiskNormal. The RiskTriang function is specified as a triangular distribution with three points which are minimum, most likely and maximum. This was considered most appropriate for the quantity used and for the spray gun opening. As an example, the most likely, the minimum and the maximum are 100, 50 and 150 mL of quantity used, respectively. The GM and GSD of LEV workplace protection factor values were entered in the RiskLognorm distribution. The air changes per hour were assumed to be normal distribution and entered in the RiskNormal distribution. The RiskTruncate function was used to truncate the inputs distribution functions within the minimum and maximum values. All these figures were entered into the exposure model as in Eq. 5 (Cherrie and Schneider, 1999):
| (5) |
In the MC simulation, the maximum concentrations and concentrations with 95% certainty were estimated. The workplace mean concentration was calculated from the personal monitoring results of ventilation on and off. These results were determined from the probability distributions.
R-squared value determines the regression values with multiple input values which is explained by the linear relationship. The linear regression is the relationship between the inputs and outputs if the number is more than 60%.
Workplace selected: Seven companies were selected for assessment based on the efficiency of their LEV: a chemistry lab (1), a paint facility (1), Motor Vehicle Repair (MVR) company (2), three sites in the wood industry (3) and one welding facility (1). All industries had different types of LEVs: three fume hood cupboard units (diethyl ether), three spray booth units (xylene), one movable LEV and one partial mixing hood unit (xylene), three movable welding hoods (welding fumes) and three radial arm saw units (wood dust).
Average face velocity results: All the face velocity results were within the limit of appropriate standard.
Workplace protection factor results: The geometric mean of WPF for different types of LEV ranged over various processes.
Sensitivity analysis of solvents and particulates: A sensitivity analysis of MC simulation indicated which inputs have the greatest contribution in influencing the outputs. The negative values are the most effective in reducing concentration exposure at the workplace, whereas a positive value increases the concentration exposure level.
Workplace protection factors for LEV systems: The aim of this research was to determine the typical protection afforded by a variety of LEV systems and to establish a system of WPF for LEV systems similar to that used for Respiratory Protective Equipment (RPE) or Hearing Protection Devices (HPD). In the work for this thesis personal monitoring was conducted with and without LEV during a range of tasks in order to calculate the WPF for different types of LEV systems.
The WPF of an LEVs can also be used to determine the estimated Maximum Use Concentration (MUC), calculated as the OEL multiplied by the WPF of the LEV as Eq. 6. If LEVs is not available, the most suitable respirator can be selected based on the MUC. In this case the respirator acts as a temporary protection equipment.
| (6) |
WPF variability -probabilistic modeling: The variability in exposure levels measured with the LEV systems on and off were influenced by several factors such as inter and intra worker variation in terms of how the task was performed, changes in environmental conditions and differences in the effectiveness of the LEV systems. For this study we have assumed that the variation that had arisen solely depended on the effectiveness of the LEV systems. While this is a gross over-simplification of the real-life situation, it has enabled us to consider the ‘worst case’ situation for each LEV system.
The Geometric Mean (GM) and Geometric Standard Deviation (GSD) of the WPF were entered into retrospective exposure model using the @RISK software to perform MC simulation. In the MC simulation, the mean, the maximum and 95th percentile concentration figures were calculated in order to estimate the likely wrst case and 95th percentile exposure levels in the workplace.
Many variables are likely to influence the exposure level, for instance, the length of the handling period, the quantity of substances used, task handling practices, air movements, equipment handling practices and the distance between the hood and the source. These variables were used as inputs in estimating the exposure concentration in the Monte Carlo simulation by using the Cherrie model (Cherrie and Schneider, 1999).
The quantity of substances used, task handling practices, air movements, equipment handling practices and WPF inputs have been entered in the Monte Carlo (MC) simulations to produce the exposure concentration. In these cases, the solvents were diethyl ether and xylene concentrations. The particulates were welding fumes and wood dust concentrations. The MC simulations determined the model parameters with the greatest impact on the estimated exposure level using sensitivity analyses techniques.
Overall, the objectives of this research have been achieved. The efficacy of a range of LEVs was determined across a number of workplaces. The WPF has been developed for different types of LEV in various processes. The 5th percentile figure can be used as protection guidance for various processes especially when LEVs are not available and to decide on the utilization of RPE at the workplace as explained in Section 6.1 (Eq. 6). The 5th percentile will overestimate exposure in 95% of cases but if control is based on this, then, only 5% of workers will not be protected. This consideration is especially important if the contaminant is highly toxic in nature.
Further study is required to produce the WPF of LEV in different tasks and for various durations of exposure. The MC simulation showed that ACH and LEV were the greatest influences on the inputs in reducing the exposure concentration. The MC simulation also enabled a determination of the uncertainty of the variables in the retrospective exposure assessment model.
The author would like to thank Professor Jon Ayres and Dr Sean Semple (Department of Environmental and Occupational Medicine, University of Aberdeen) and Dr Kausar Ahmad (Department of Pharmaceutical Technology, International Islamic University Malaysia) for their assistance, guidance and patience and thank you to his family and friends for their support and understanding, which have assisted him in completing the thesis.