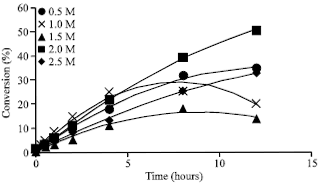
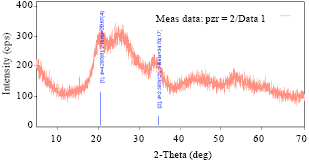
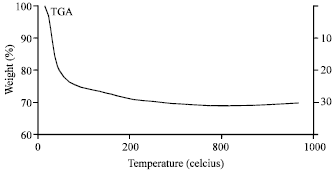
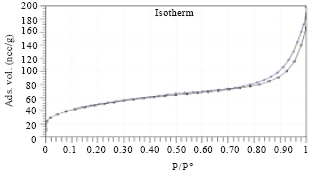
Research Article
Synthesis and Characterization of Zirconium Phosphate as a Solid Catalyst for Esterification of Wastewater Containing Acrylic Acid
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, Lebuhraya Tun Razak, 26300 Kuantan, Pahang, Malaysia
S.Y. Chin
Faculty of Chemical and Natural Resources Engineering, Universiti Malaysia Pahang, Lebuhraya Tun Razak, 26300 Kuantan, Pahang, Malaysia