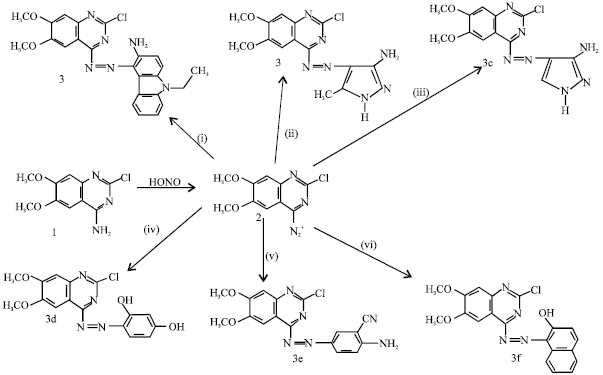
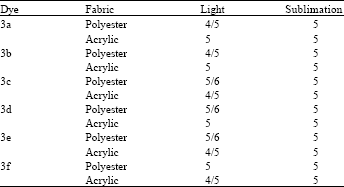
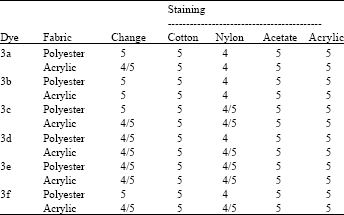
Research Article
Synthesis of Heterocyclic Monoazo Disperse Dyes Derived from 4-Amino-2-chloro-6, 7-dimethoxyquinazoline
Department of Chemistry, Faculty of Science, Delta State University, P.M.B. 1, Abraka, Delta State, Nigeria
E.M. Efurhievwe
Nigerian National Petroleum Company, NAPIMS, JV Operations, Well Operations and Monitoring (WO/M) Gerrard Road, Ikoyi Lagos, Nigeria