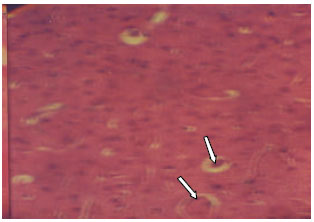
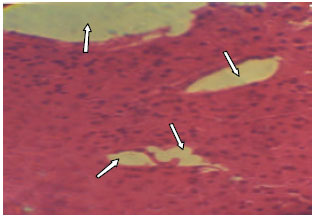
Research Article
Effect of L-Arginine on Some Biochemical Markers of Metabolic Syndrome Associated with Brain Function in Female Wistar Rats
Department of Biochemistry, University of Nigeria, Nsukk, Enugu State, Nigeria
L.U.S. Ezeanyika
Department of Biochemistry, University of Nigeria, Nsukk, Enugu State, Nigeria