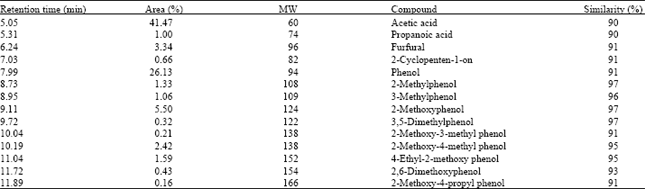
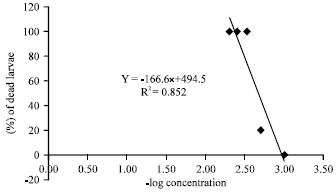
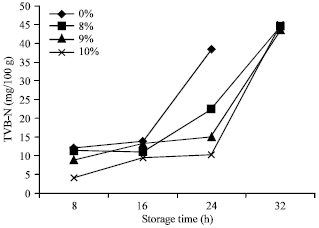
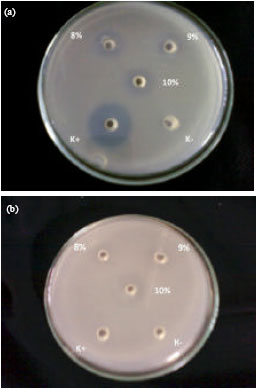
Research Article
Characterization of Redistilled Liquid Smoke of Oil-palm Shells and its Application as Fish Preservatives
Department of Chemistry, Bogor Agricultural University, Kampus Dramaga, Bogor 16680, Indonesia
N.R. Mubarik
Department of Biology, Bogor Agricultural University, Kampus Dramaga, Bogor 16680, Indonesia
R. Nursyamsi
Department of Chemistry, Bogor Agricultural University, Kampus Dramaga, Bogor 16680, Indonesia
P. Septiaji
Department of Chemistry, Bogor Agricultural University, Kampus Dramaga, Bogor 16680, Indonesia