Research Article
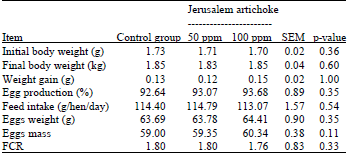
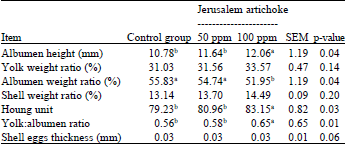
Effect of Jerusalem Artichoke (Helianthus tuberosus L.) Supplementation on Production Performances, Egg Quality Characteristics and Intestinal Microflora of Laying Hens
Department of Animal Science, Faculty of Agriculture, Kasetsart University, Bangkok
C. Kaewtapee
Department of Animal Science, Faculty of Agriculture, Kasetsart University, Bangkok
C. Bunchasak
Department of Animal Science, Faculty of Agriculture, Kasetsart University, Bangkok
T. Poeikhampha
Department of Animal Science, Faculty of Agriculture, Kasetsart University, Bangkok