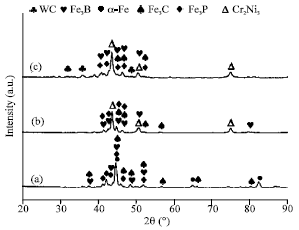
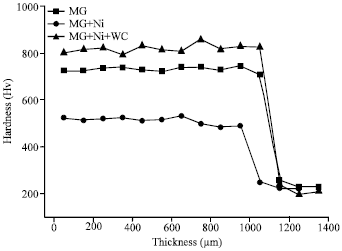
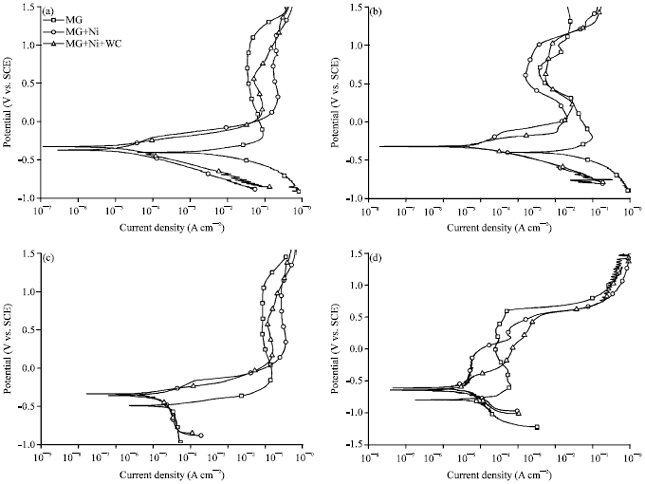
Research Article
Corrosion Resistance of Coating with Fe-based Metallic Glass Powders Fabricated by Laser Spraying
Power Equipment Factory of Jiangxi, Nanchang, 330100, China
Ouyang Gengsheng
Power Equipment Factory of Jiangxi, Nanchang, 330100, China
Zhang Lei
Power Equipment Factory of Jiangxi, Nanchang, 330100, China
Wu Xiaoping
Power Equipment Factory of Jiangxi, Nanchang, 330100, China
Zhang Hengyu
School of Aeronautic Manufacturing Engineering, Nanchang Hangkong University, Nanchang, 330063, China
Wang Shanlin
School of Aeronautic Manufacturing Engineering, Nanchang Hangkong University, Nanchang, 330063, China