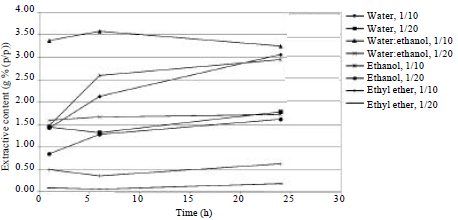
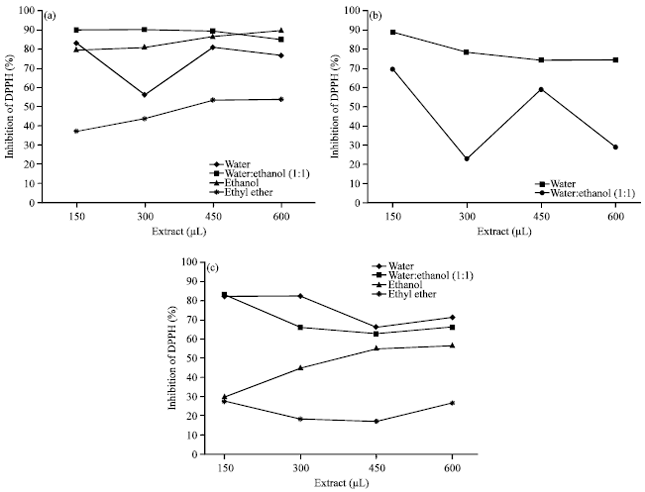
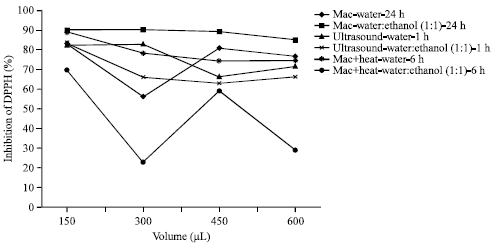
Research Article
Optimization of Extraction Conditions and Antioxidant Activity of Solanum lycocarpum Fruits
University Center of the Educational Foundation of Barretos, Barretos, S�o Paulo State, Brazil
A.N. dos Santos
University Center of the Educational Foundation of Barretos, Barretos, S�o Paulo State, Brazil
M.V. Ribeiro Vilela
University Center of the Educational Foundation of Barretos, Barretos, S�o Paulo State, Brazil
M. de Carvalho Ferreira
University Center of the Educational Foundation of Barretos, Barretos, S�o Paulo State, Brazil
W. Pereira de Oliveira
Faculty of Pharmaceutical Sciences of Ribeirao Preto, University of Sao Paulo, Ribeir�o Preto, S�o Paulo State, Brazil
R.R. Duarte Moreira
Department Natural Active Principles and Toxicology, Faculty of Pharmaceutical Sciences, State University Paulista, Araraquara, S�o Paulo State, Brazil