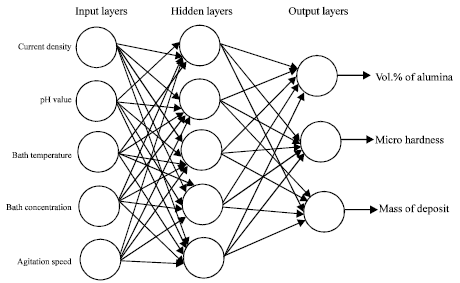
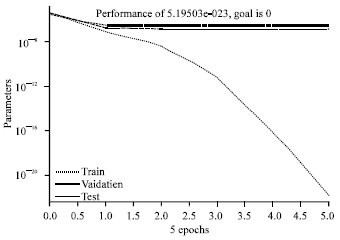
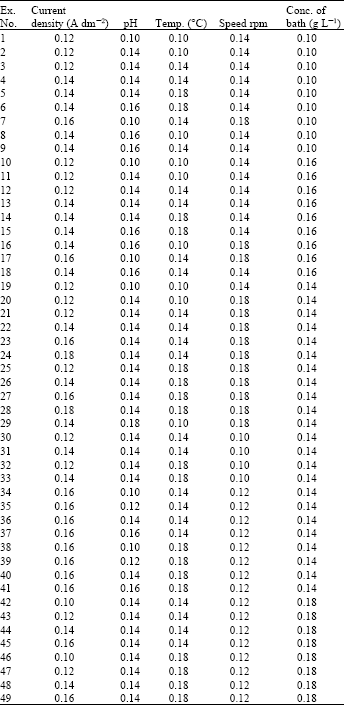
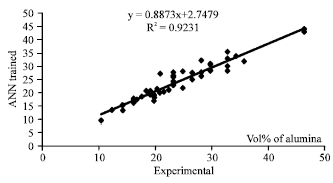
Research Article
Characterization of Electrodeposited Nickel-Al2O3 Composite Coatings by Experimental Method and Neural Networks
School of Mechanical Engineering, SASTRA University, Thanjavur, 613401, India
K.P. Arulshri
Principal, K.P.R. Institute of Engineering and Technology, Coimbatore, 641407, India